270253
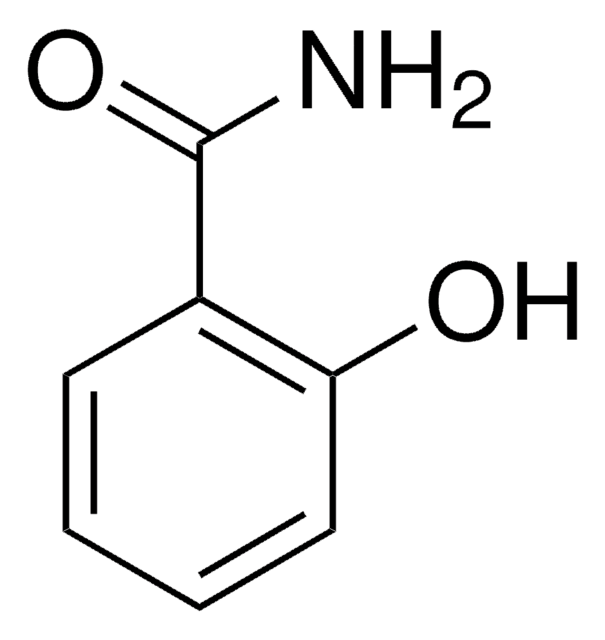
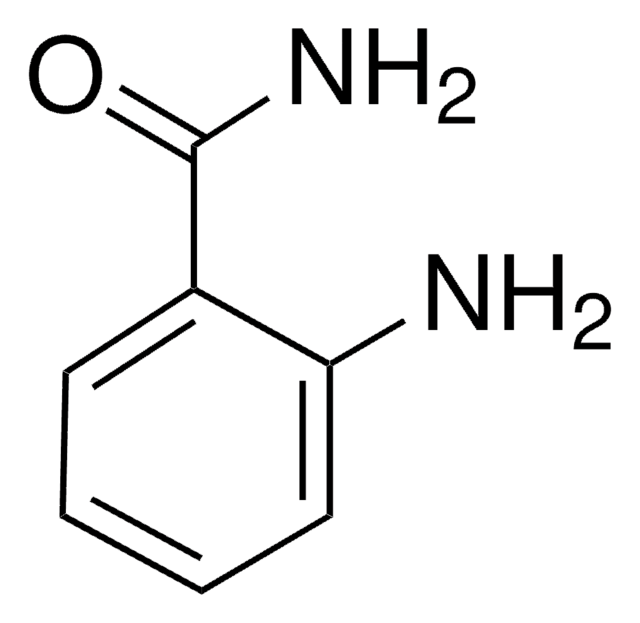
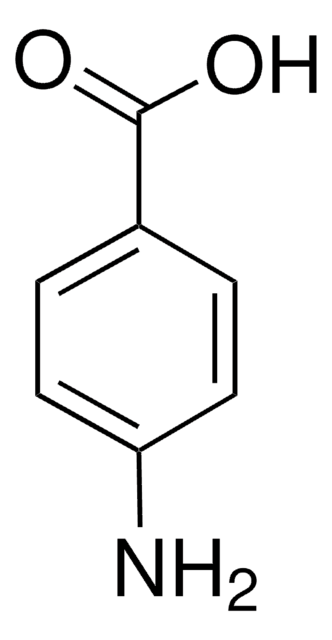
4-Hydroxybenzamide
98%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
HOC6H4CONH2
Numero CAS:
Peso molecolare:
137.14
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Punto di fusione
161-162 °C (lit.)
Gruppo funzionale
amide
Stringa SMILE
NC(=O)c1ccc(O)cc1
InChI
1S/C7H7NO2/c8-7(10)5-1-3-6(9)4-2-5/h1-4,9H,(H2,8,10)
QXSAKPUBHTZHKW-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
The standard molar enthalpy of formation of 4-hydroxybenzamide was studied by micro- or macrocombustion calorimetry.
Applicazioni
4-Hydroxybenzamide was used in the synthesis of balanol, a potent protein kinase C (PKC) inhibitor.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
R S Randad et al.
Bioorganic & medicinal chemistry, 4(9), 1471-1480 (1996-09-01)
A combination of structure-activity studies, kinetic analysis, X-ray crystallographic analysis, and modeling were employed in the design of a novel series of HIV-1 protease (HIV PR) inhibitors. The crystal structure of a complex of HIV PR with SRSS-2,5-bis[N-(tert-butyloxycarbonyl)amino]-3,4-dihydroxy-1, 6-diphenylhexane (1)
G D Hartman et al.
Bioorganic & medicinal chemistry letters, 9(6), 863-868 (1999-04-17)
A new series of potent, linearly-minimized, orally active, selective GPIIb/IIIa inhibitors is identified. Thus 15 (L-750,034) achieves interaction via a constrained, non-turned conformation that maintains the proper distance between its charged termini and full sulfonamide exosite interaction. The diminutive stature
C Emoto et al.
Xenobiotica; the fate of foreign compounds in biological systems, 37(12), 1408-1420 (2007-10-19)
CJ-036878, N-(3-phenethoxybenzyl)-4-hydroxybenzamide, was developed as an antagonist of the N-methyl-D-aspartate receptor NR2B subunit. Two dimeric metabolites, CJ-047710 and CJ-047713, were identified from the incubation mixture with CJ-036878 in human liver microsomes (HLM). The identification of the enzymes involved in the
Carlos E S Bernardes et al.
The journal of physical chemistry. A, 112(40), 10029-10039 (2008-09-13)
The energetics of the phenolic O-H bond in a series of 2- and 4-HOC 6H 4C(O)Y (Y = H, CH3, CH 2CH=CH2, C[triple bond]CH, CH2F, NH2, NHCH 3, NO2, OH, OCH3, OCN, CN, F, Cl, SH, and SCH3) compounds and
G D Smith et al.
Protein science : a publication of the Protein Society, 5(8), 1502-1511 (1996-08-01)
The structure of a symmetric T3R3f insulin hexamer, complexed with 4-hydroxybenzamide, has been determined using X-ray crystallographic techniques. Data were measured from six crystals grown in microgravity to a resolution of 1.4 A and the structure has been refined including
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.