108057
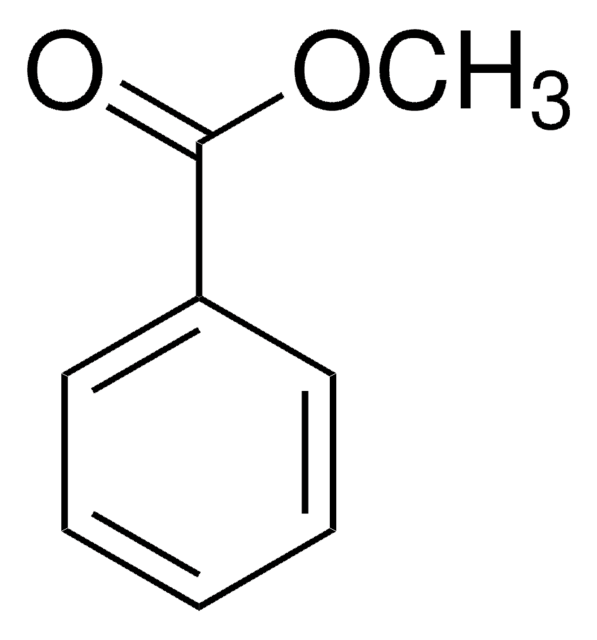
Methyl phenylacetate
ReagentPlus®, ≥99%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C6H5CH2CO2CH3
Numero CAS:
Peso molecolare:
150.17
Beilstein:
878795
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Nome Commerciale
ReagentPlus®
Saggio
≥99%
Indice di rifrazione
n20/D 1.503 (lit.)
P. ebollizione
218 °C (lit.)
Densità
1.066 g/mL at 20 °C (lit.)
Stringa SMILE
COC(=O)Cc1ccccc1
InChI
1S/C9H10O2/c1-11-9(10)7-8-5-3-2-4-6-8/h2-6H,7H2,1H3
CRZQGDNQQAALAY-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
Methyl phenylacetate was used as model compound for partition coefficient measurement experiments.
Azioni biochim/fisiol
Methyl phenylacetate undergoes decomposition on photolysis in methanol. Methyl phenylacetate acts as acylating agent and causes the enantioselective acylation of beta-lactam intermediate using penicillin G amidase. Methyl phenylacetate is the starting material in manufacture of synthetic perfumes.
Note legali
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Codice della classe di stoccaggio
10 - Combustible liquids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
211.1 °F - closed cup
Punto d’infiammabilità (°C)
99.5 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Photolysis of phenylacetic acid and methyl phenylacetate in methanol.
Meiggs TO and Miller SI.
Journal of the American Chemical Society, 94(6), 1989-1996 (1972)
Yichen Cao et al.
Journal of pharmaceutical sciences, 93(11), 2768-2779 (2004-09-25)
The ability to predict drug solubility and partitioning in triglyceride solvents from the chemical structures of the solute and the triglyceride would be highly useful in drug formulation development and in screening drug candidates for lipid solubility and possibly drug
Jonathan Slaughter et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 23(1), 167-175 (2016-10-30)
In investigating and seeking to mimic the reactivity of trimethylaluminium (TMA) with synthetic, ester-based lubricating oils, the reaction of methyl propionate 1 was explored with 1, 2 and 3 equivalents of the organoaluminium reagent. Spectroscopic analysis points to the formation of
Enantioselective acylation of a beta-lactam intermediate in the synthesis of loracarbef using penicillin G amidase.
Zmijewski Jr MJ, et al.
Tetrahedron Letters, 32(13), 1621-1622 (1991)
Miriam Frida Karlsson et al.
Journal of agricultural and food chemistry, 57(13), 5903-5909 (2009-06-06)
The Guatemalan moth Tecia solanivora is an invasive pest of potato in Central and South America. The larvae infest potato tubers in the field as well as in storage facilities. The headspace of potato foliage and potato tubers was studied
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.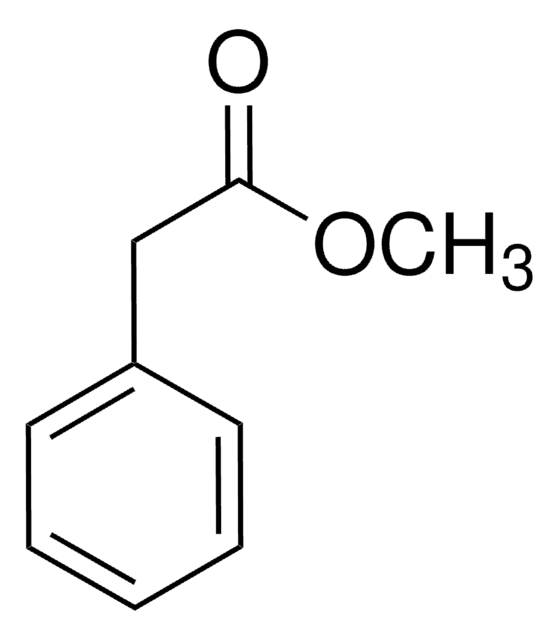



![1,8-diazabiciclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)