287512
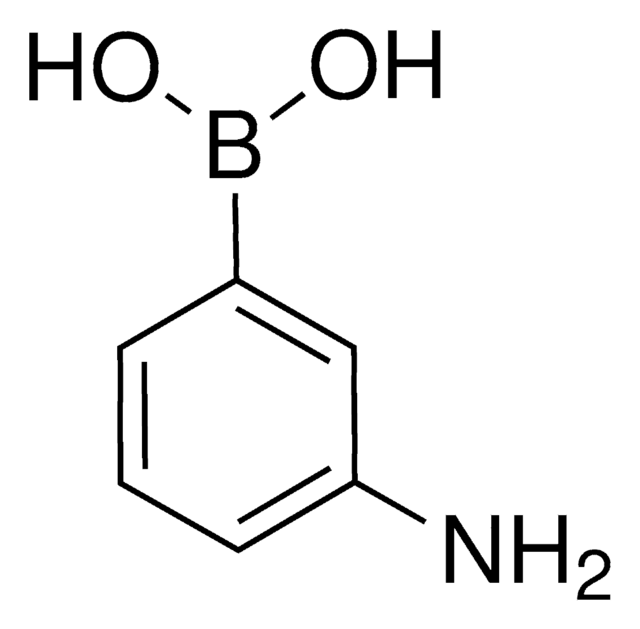
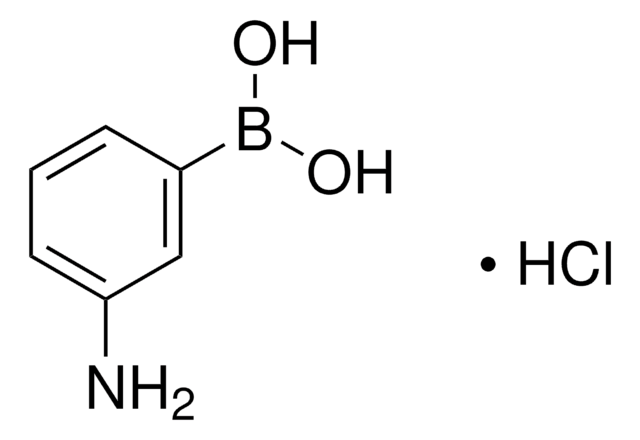
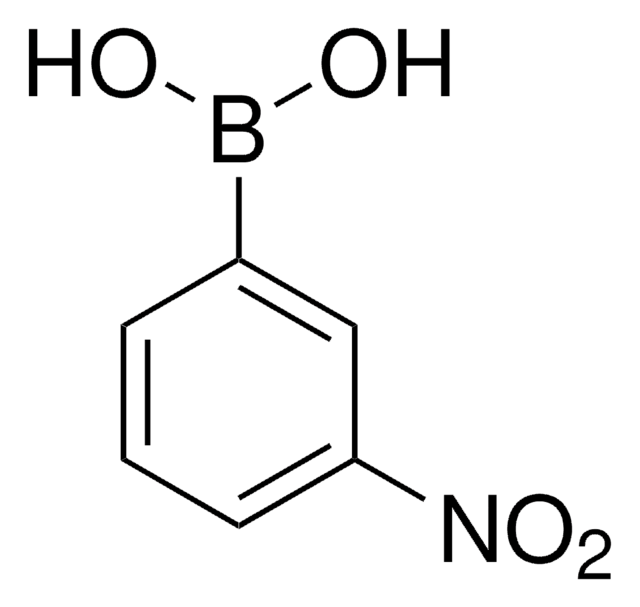
3-Aminophenylboronic acid monohydrate
98%
Synonym(s):
3-Aminobenzeneboronic acid
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
H2NC6H4B(OH)2 · H2O
CAS Number:
Molecular Weight:
154.96
Beilstein:
2936342
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
mp
93-96 °C (lit.)
SMILES string
[H]O[H].Nc1cccc(c1)B(O)O
InChI
1S/C6H8BNO2.H2O/c8-6-3-1-2-5(4-6)7(9)10;/h1-4,9-10H,8H2;1H2
InChI key
XAEOVQODHLLNKX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Reagent used for
Reagent used for Preparation of
- Suzuki-Miyaura cross-coupling
Reagent used for Preparation of
- Gram-positive antivirulence drugs and inhibitors of Streptococcus agalactiae Stk1
- Regioisomer of Zaleplon (a sedative)
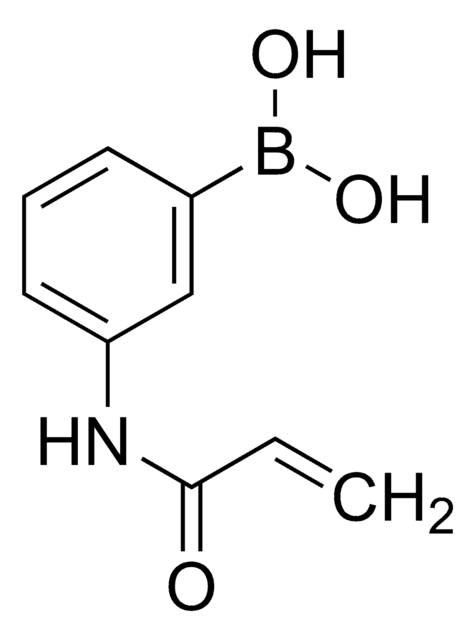
- Amphiphilic random glycopolymer, which self-assemble to form nanoparticles, with potential as a glucose-sensitive matrix
- Chemomechanical polymer that expands and contracts in blood plasma with high glucose selectivity
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Jing Zhang et al.
Journal of separation science, 40(5), 1107-1114 (2017-01-04)
Novel 3-aminophenylboronic acid functionalized poly(glycidyl methacrylate-co-ethylene dimethacrylate) microspheres were prepared for the solid-phase extraction of glycopeptides/glycoproteins. The adsorption efficiency, maximum adsorption capacity, and specific recognition of the microspheres to glycoprotein were investigated. The results indicated excellent adsorption of glycoproteins by
Gizem Ertürk Bergdahl et al.
Applied biochemistry and biotechnology, 189(2), 374-383 (2019-04-26)
A capacitive sensor was developed to analyze the presence and enzymatic activity of a model protease from standard solutions by following the degradation of the substrate in real time. The enzyme was chosen based on its specific digestion of the
Aishwarya Mahadevan et al.
Biosensors & bioelectronics, 92, 417-424 (2016-11-12)
An improved glycerol biosensor was developed via direct attachment of NAD
Mayalen Oxoby et al.
Bioorganic & medicinal chemistry letters, 20(12), 3486-3490 (2010-06-10)
A structure-activity relationship study from a screening hit and structure-based design strategy has led to the identification of bisarylureas as potent inhibitors of Streptococcus agalactiae Stk1. As this target has been directly linked to bacterial virulence, these inhibitors can be
Xingju Jin et al.
Biomacromolecules, 10(6), 1337-1345 (2009-04-29)
This study is devoted to developing amphiphilic, random glycopolymers based on phenylboronic acid, which self-assemble to form nanoparticles (NPs), as a glucose-sensitive agent. Maleimide-glucosamine was copolymerized with 3-acryl aminophenylboronic acid in methanol at 70 degrees C. Using the nanoprecipitation method
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service