349801
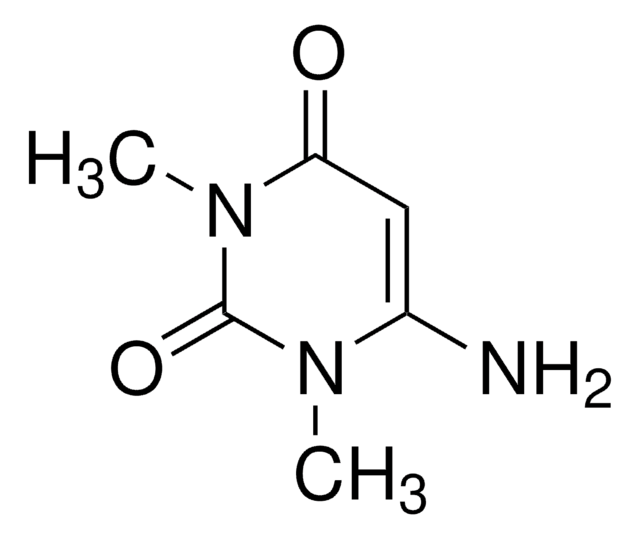
1,3-Dimethyluracil
99%
Synonym(s):
1,3-Dimethyl-2,4(1H,3H)-pyrimidinedione, 2,4-Dihydroxy-1,3-dimethylpyrimidine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H8N2O2
CAS Number:
Molecular Weight:
140.14
Beilstein:
124074
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
powder
mp
119-122 °C (lit.)
SMILES string
CN1C=CC(=O)N(C)C1=O
InChI
1S/C6H8N2O2/c1-7-4-3-5(9)8(2)6(7)10/h3-4H,1-2H3
InChI key
JSDBKAHWADVXFU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
1,3-Dimethyluracil is a pyrimidine derivative. Stability of the C6-centered carbanions derived from 1,3-dimethyluracil has been investigated in the gas phase and in DMSO and water solutions. The excited state structural dynamics of 1,3-dimethyluracil (DMU) in water and acetonitrile has been studied by resonance Raman spectroscopy. Crystal structure of 1,3-dimethyluracil has been reported. Ultraviolet irradiation of aqueous 1,3-dimethyluracil results in hydration of the 5:6 double bond of the uracil ring to form 1,3-dimethyl-6-oxy-hydrouracil.
Application
1,3-Dimethyluracil is suitable reagent used to investigate the steady-state absorption and fluorescence spectra of uracil derivatives. It may be used in the preparation of 2,6-dihydroxynicotinamide.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Adam Gryff-Keller et al.
The journal of physical chemistry. A, 116(39), 9632-9638 (2012-09-14)
The practical utility of the method of retrieving the relaxation rate of a quadrupole nucleus via the scalar relaxation of the second kind (SC2) of an I = 1/2 spin nucleus has been considered once again. The study was motivated
Nicholas A Senger et al.
Tetrahedron, 69(26), 5287-5292 (2013-09-28)
The stabilities of the C6-centered carbanions derived from 1,3-dimethyluracil, N-methyl-2-pyridone, and N-methyl-4-pyridone were systematically investigated in the gas phase and in DMSO and water solutions. The stabilities of the carbanions in the gas phase and DMSO were directly measured through
Probing noncovalent interactions in biomolecular crystals with terahertz spectroscopy.
Thomas Kleine-Ostmann et al.
Chemphyschem : a European journal of chemical physics and physical chemistry, 9(4), 544-547 (2008-02-15)
H P Schuchmann et al.
International journal of radiation biology and related studies in physics, chemistry, and medicine, 50(6), 1051-1068 (1986-12-01)
Hydroxymethyl radicals .CH2OH, generated by the radiolysis of methanol (0.5 mol dm-3) in N2O-saturated aqueous solutions, were reacted with 1,3-dimethyluracil or 1,3-dimethylthymine (10(-3) mol dm-3). The products were identified and their G values determined. It has been concluded that in
Anna A Zadorozhnaya et al.
The journal of physical chemistry. A, 114(4), 2001-2009 (2010-01-09)
The electronic structure of 1,3-dimethyluracil and its dimer is characterized by ab initio calculations. The methylation eliminates the H-bonded isomers and allows one to focus on the pi-stacked manifold. In the neutral species, methylation increases the binding energy by 3-4
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service