SMB00912
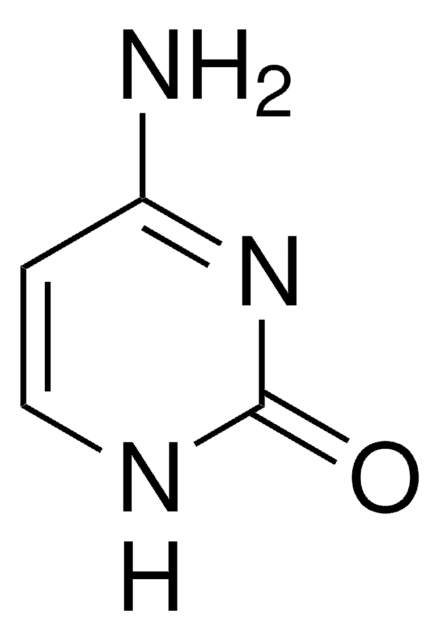
Pseudouridine
≥98% (HPLC)
Synonym(s):
Pseudouridine, β-Pseudouridine, ψ-Uridine, 5-(β-D-Ribofuranosyl)uracil
Sign Into View Organizational & Contract Pricing
Select a Size
All Photos(1)
Select a Size
Change View
About This Item
Empirical Formula (Hill Notation):
C9H12N2O6
CAS Number:
Molecular Weight:
244.20
UNSPSC Code:
41106305
NACRES:
NA.79
Recommended Products
biological source
synthetic (chemical)
Quality Level
Assay
≥98% (HPLC)
form
powder
mol wt
244.2
color
white to off-white
mp
222 °C ((432 °F ))
solubility
water: soluble
storage temp.
2-8°C
SMILES string
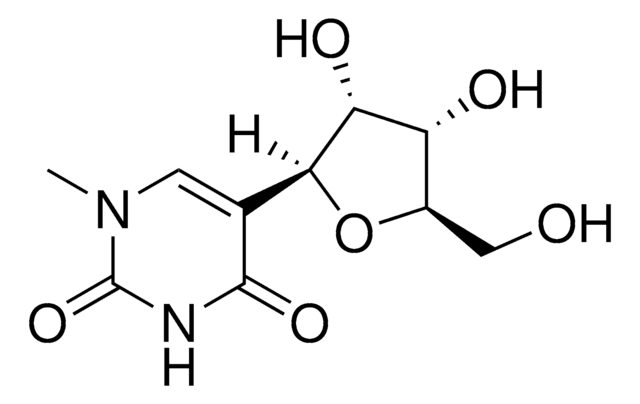
[nH]1[c]([nH]cc([c]1=O)[C@@H]2O[C@@H]([C@H]([C@H]2O)O)CO)=O
General description
Pseudouridine (Ψ) stands as an isomer of the nucleoside uridine, featuring a carbon-carbon bond instead of the typical nitrogen-carbon glycosidic bond connecting uracil. It represents the most prevalent among the myriad modified nucleosides within RNA, with a presence spanning various species and RNA classes. The enzymatic action of Ψ synthases induces post-transcriptional isomerization of specific uridine residues, a process known as pseudouridylation. This modification, particularly in rRNA and tRNA, plays a vital role in fine-tuning and stabilizing regional structures, contributing to mRNA decoding, ribosome assembly, processing, and translation functions. Moreover, β-pseudouridine, identified in tRNAs across bacteria, archaea, and eukaryotes, has demonstrated potential in reducing radiation-induced chromosome aberrations in human lymphocytes. Its utility as a cancer and proliferation biomarker positions it as a valuable asset in metabolomics and biochemical research.
Pseudouridine is a C-glycosyl pyrimidine that consists of uracil having a β-D-ribofuranosyl residue attached at position 5. The C-glycosyl isomer of the nucleoside uridine. It has a role as a fundamental metabolite. Pseudouridine is found in all species and in all classes of RNA except mRNA. It is formed by enzymes called pseudouridine synthases, which post-transcriptionally isomerize specific uridine residues in RNA.
Application
Pseudouridine is a versatile compound and a biomarker that finds application in metabolomics and biochemical research.
Features and Benefits
- High-purity compound suitable for a wide variety of research applications
Other Notes
For additional information on our range of Biochemicals, please complete this form.
To gain a comprehensive understanding of our extensive range of Monosaccharides for your research, we encourage you to visit our Carbohydrates Category page.
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Mohammad Ariful Islam et al.
Biomaterials, 266, 120431-120431 (2020-10-26)
Synthetic mRNA represents an exciting cancer vaccine technology for the implementation of effective cancer immunotherapy. However, inefficient in vivo mRNA delivery along with a requirement for immune co-stimulation present major hurdles to achieving anti-tumor therapeutic efficacy. Here, we demonstrate a
Ryota Kurimoto et al.
The EMBO journal, 39(20), e104708-e104708 (2020-09-15)
Let-7 is an evolutionary conserved microRNA that mediates post-transcriptional gene silencing to regulate a wide range of biological processes, including development, differentiation, and tumor suppression. Let-7 biogenesis is tightly regulated by several RNA-binding proteins, including Lin28A/B, which represses let-7 maturation.
Matthias Heiss et al.
Nature communications, 12(1), 389-389 (2021-01-17)
Recently, studies about RNA modification dynamics in human RNAs are among the most controversially discussed. As a main reason, we identified the unavailability of a technique which allows the investigation of the temporal processing of RNA transcripts. Here, we present
Tingting Jiang et al.
Nature communications, 11(1), 1979-1979 (2020-04-26)
CRISPR-Cas9-associated base editing is a promising tool to correct pathogenic single nucleotide mutations in research or therapeutic settings. Efficient base editing requires cellular exposure to levels of base editors that can be difficult to attain in hard-to-transfect cells or in
Tatsuro Hagi et al.
Applied microbiology and biotechnology, 104(24), 10641-10653 (2020-11-08)
Akkermansia muciniphila is a prominent member of the gut microbiota and the organism gets exposed to bile acids within this niche. Several gut bacteria have bile response genes to metabolize bile acids or an ability to change their membrane structure
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service