05512
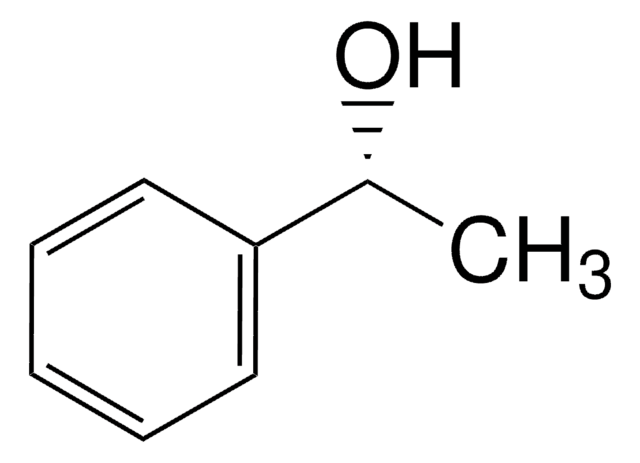
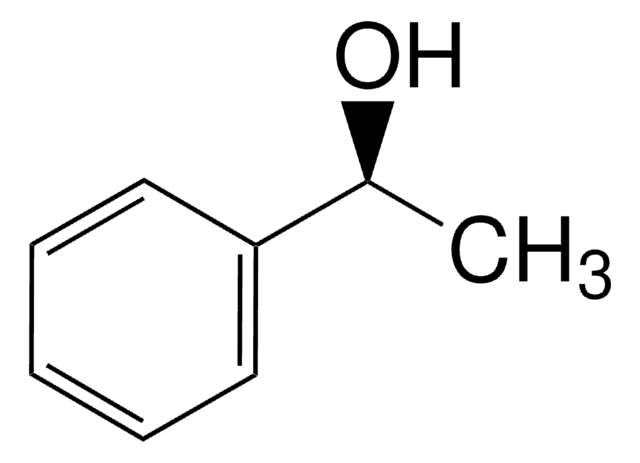
(S)-(−)-1-Phenylethanol
≥98.5% (sum of enantiomers, GC)
Synonym(s):
(−)-Methyl phenyl carbinol, (S)-(−)-α-Methylbenzyl alcohol, (S)-(−)-sec-Phenylethyl alcohol
About This Item
Recommended Products
Quality Level
Assay
≥98.5% (sum of enantiomers, GC)
form
liquid
optical activity
[α]/D −45±2°, c = 5% in methanol
optical purity
enantiomeric ratio: ≥97:3 (GC)
refractive index
n20/D 1.527
bp
88-89 °C/10 mmHg (lit.)
mp
9-11 °C (lit.)
density
1.012 g/mL at 20 °C (lit.)
functional group
hydroxyl
phenyl
SMILES string
C[C@H](O)c1ccccc1
InChI
1S/C8H10O/c1-7(9)8-5-3-2-4-6-8/h2-7,9H,1H3/t7-/m0/s1
InChI key
WAPNOHKVXSQRPX-ZETCQYMHSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- A starting material to prepare (1S,3R,4S)-1-methyl-3,4-diphenyl-3,4-dihydro-1H-isochromene-3,4-diol (a cyclic hemiacetal) by reacting with benzil via dilithiation reaction.
- A chiral solvent in the symmetric synthesis of substituted spiroundecenetriones via amino acid-catalyzed domino Knoevenagel/Diels-Alder reactions.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
WGK
WGK 3
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Chiral Alcohols
Chromatograms
suitable for GCOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service