推荐产品
等級
pharmaceutical primary standard
API 家族
irbesartan
製造商/商標名
USP
應用
pharmaceutical (small molecule)
格式
neat
SMILES 字串
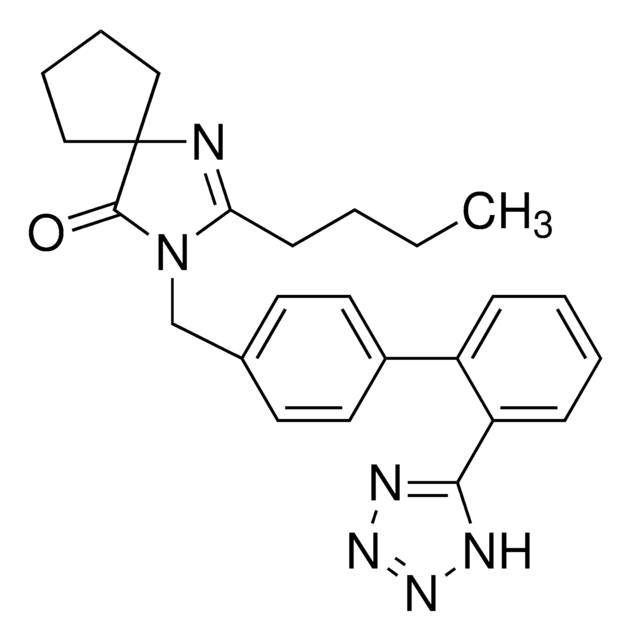
CCCCC1=NC2(CCCC2)C(=O)N1Cc3ccc(cc3)-c4ccccc4-c5nnn[nH]5
InChI
1S/C25H28N6O/c1-2-3-10-22-26-25(15-6-7-16-25)24(32)31(22)17-18-11-13-19(14-12-18)20-8-4-5-9-21(20)23-27-29-30-28-23/h4-5,8-9,11-14H,2-3,6-7,10,15-17H2,1H3,(H,27,28,29,30)
InChI 密鑰
YOSHYTLCDANDAN-UHFFFAOYSA-N
基因資訊
human ... AGTR1(185)
正在寻找类似产品? 访问 产品对比指南
相关类别
應用
Irbesartan USP Reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monographs such as:
- Irbesartan Tablets
- Irbesartan and Hydrochlorothiazide Tablets
分析報告
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
其他說明
Sales restrictions may apply.
相關產品
产品编号
说明
价格
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
J C Gillis et al.
Drugs, 54(6), 885-902 (1998-01-09)
Irbesartan inhibits the activity of angiotensin II (AII) via specific, selective noncompetitive antagonism of the AII receptor subtype 1 (AT1) which mediates most of the known physiological activities of AII. In patients with mild to moderate hypertension, once daily administration
Optimizing antiplatelet therapy in high-risk patients with atrial fibrillation: insights from Atrial Fibrillation Clopidogrel Trial with Irbesartan for Prevention of Vascular Events (ACTIVE A).
Raymond Chee-Seong Seet et al.
Stroke, 40(12), 3883-3885 (2009-10-31)
Katherine F Croom et al.
Drugs, 68(11), 1543-1569 (2008-07-17)
Irbesartan (Aprovel, Avapro, Irbetan, Karvea), an angiotensin II receptor type 1 antagonist, is approved in many countries worldwide for the treatment of hypertension. It is also approved in some regions for the treatment of nephropathy in patients with hypertension and
Andrea Hartner et al.
Biochimica et biophysica acta, 1842(4), 558-565 (2014-01-15)
Diabetes can disrupt endoplasmic reticulum (ER) homeostasis which leads to ER stress. ER stress-induced renal apoptosis seems to be involved in the development of diabetic nephropathy. The present study was designed to investigate the contribution of reduced ER stress to
Karly P Garnock-Jones
American journal of cardiovascular drugs : drugs, devices, and other interventions, 13(2), 141-150 (2013-03-22)
Combination therapy is often required in patients with hypertension, and fixed-dose single-pill combinations have been shown to provide an easier regimen for patients, improving adherence. Irbesartan/amlodipine (Aprovasc®) is an angiotensin-receptor blocker/calcium-channel blocker fixed-dose single-pill combination, whose constituent drugs exert additive
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门