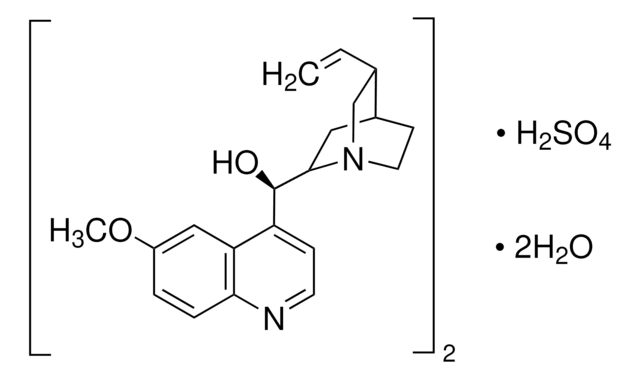
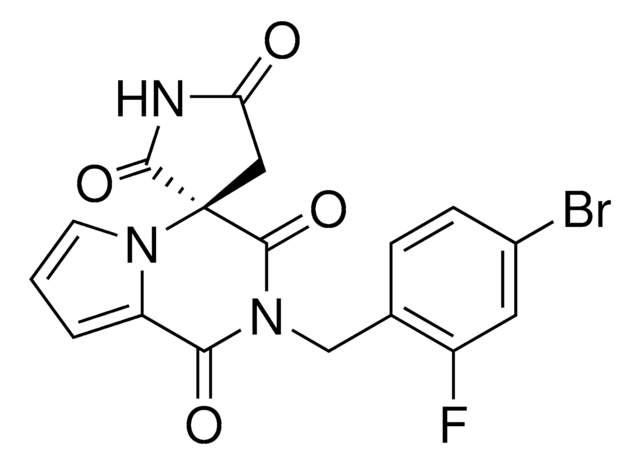
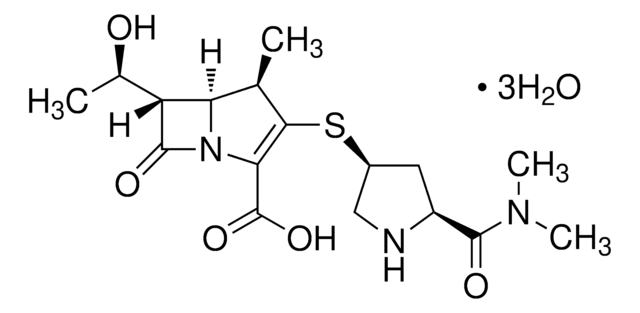
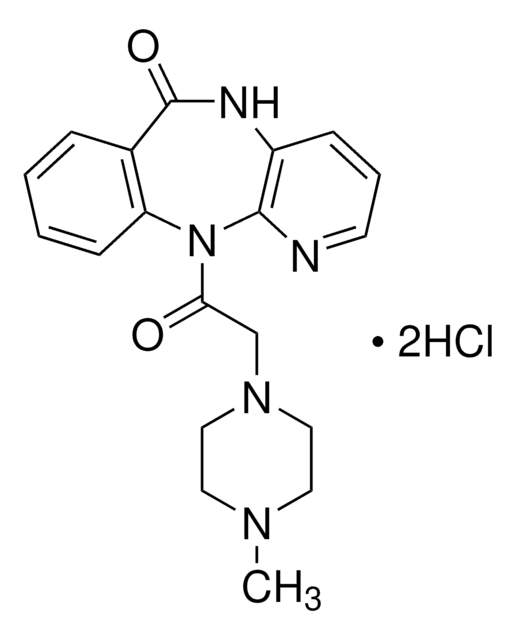
推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
powder
光學活性
[α]/D +50 to +60°, c = 1 in methanol
顏色
white to off-white
溶解度
DMSO: ≥20 mg/mL
儲存溫度
2-8°C
SMILES 字串
Fc1ccc2OCC[C@]3(NC(=O)NC3=O)c2c1
InChI
1S/C11H9FN2O3/c12-6-1-2-8-7(5-6)11(3-4-17-8)9(15)13-10(16)14-11/h1-2,5H,3-4H2,(H2,13,14,15,16)/t11-/m0/s1
InChI 密鑰
LXANPKRCLVQAOG-NSHDSACASA-N
應用
Sorbinil has been used as an aldose reductase inhibitor in diabetic rats as a positive control group.
Sorbinil may be used in cell signaling and neuroscience studies.
生化/生理作用
Sorbinil decreases the level of sorbitol in red blood cells and increases the velocity of nerve conduction. It maintains the myo-inositol content in the nerve and prevents the reduction of sodium-potassium ATPase activity. This action is beneficial in providing symptomatic relief in patients with diabetic neuropathy.
Sorbinil is an inhibitor of Aldose Reductase (AR). AR family members AKR1B1 and AKR1B10 have additionally been shown to play roles in inflammation and cancer.
特點和優勢
This compound is featured on the Dopamine and Norepinephrine Metabolism page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
免責聲明
RESEARCH USE ONLY. This product is regulated in France when intended to be used for scientific purposes, including for import and export activities (Article L 1211-1 paragraph 2 of the Public Health Code). The purchaser (i.e. enduser) is required to obtain an import authorization from the France Ministry of Research referred in the Article L1245-5-1 II. of Public Health Code. By ordering this product, you are confirming that you have obtained the proper import authorization.
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Oral
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Kun-Che Chang et al.
Chemico-biological interactions, 276, 149-154 (2017-02-01)
Cataract is the most frequent cause of blindness worldwide and is treated by surgical removal of the opaque lens to restore the light path to the retina. While cataract surgery is a safe procedure, some patients develop a complication of
Chiara Gerhardinger et al.
Diabetes, 58(7), 1659-1667 (2009-04-30)
Prevention of diabetic retinopathy would benefit from availability of drugs that preempt the effects of hyperglycemia on retinal vessels. We aimed to identify candidate drug targets by investigating the molecular effects of drugs that prevent retinal capillary demise in the
R J Young et al.
Diabetes, 32(10), 938-942 (1983-10-01)
A double-blind, randomized, placebo-controlled cross-over trial of the aldose reductase inhibitor sorbinil was undertaken in 15 patients (age 35-68 yr) with chronic painful diabetic neuropathy. Treatment was evaluated by subjective pain responses, clinical examination, vibration perception threshold, motor and sensory
Karin Kaiserova et al.
The Journal of biological chemistry, 283(14), 9101-9112 (2008-01-29)
Aldose reductase (AR) catalyzes the reduction of several aldehydes ranging from lipid peroxidation products to glucose. The activity of AR is increased in the ischemic heart due to oxidation of its cysteine residues, but the underlying mechanisms remain unclear. To
In vitro evaluation of 5-arylidene-2-thioxo-4-thiazolidinones active as aldose reductase inhibitors.
Rosanna Maccari et al.
Bioorganic & medicinal chemistry letters, 21(1), 200-203 (2010-12-07)
2-Thioxo-4-thiazolidinone derivatives were evaluated as aldose reductase inhibitors (ARIs) and most of them exhibited good or excellent in vitro efficacy. Out of the tested compounds, most N-unsubstituted analogues were found to possess inhibitory effects at low micromolar doses and two
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门