推荐产品
生物源
synthetic (organic)
品質等級
無菌
non-sterile
形狀
powder
溶解度
DMSO: soluble 14 mg/mL at ≤60 °C
H2O: insoluble
運輸包裝
ambient
儲存溫度
room temp
SMILES 字串
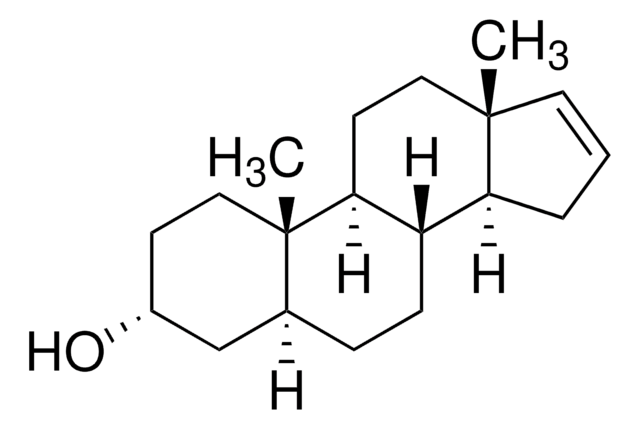
[H][C@@]12CC[C@@]3([H])[C@]4([H])CCC[C@@]4(C)CC[C@]3([H])[C@@]1(C)CC[C@H](O)C2
InChI
1S/C19H32O/c1-18-9-3-4-16(18)15-6-5-13-12-14(20)7-11-19(13,2)17(15)8-10-18/h13-17,20H,3-12H2,1-2H3/t13-,14-,15-,16-,17-,18-,19-/m0/s1
InChI 密鑰
DJTOLSNIKJIDFF-LOVVWNRFSA-N
正在寻找类似产品? 访问 产品对比指南
應用
5α-Androstan-3β-ol has been used as a cortisone analog to test its effect on the voltage-dependent potassium channel (Kv) current.
生化/生理作用
mCAR (constitutive androstane receptor) inverse agonist; testosterone metabolite.
危險聲明
防範說明
危險分類
Aquatic Chronic 4
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
A Kassam et al.
The Journal of biological chemistry, 275(6), 4345-4350 (2000-02-08)
The genes encoding the first two enzymes of the peroxisomal beta-oxidation pathway, acyl-CoA oxidase (AOx) and enoyl-CoA hydratase/3-hydroxyacyl-CoA dehydrogenase (HD), contain upstream cis-acting regulatory regions termed peroxisome proliferator response elements (PPRE). Transcription of these genes is mediated through the binding
I Tzameli et al.
Molecular and cellular biology, 20(9), 2951-2958 (2000-04-11)
A wide range of xenobiotic compounds are metabolized by cytochrome P450 (CYP) enzymes, and the genes that encode these enzymes are often induced in the presence of such compounds. Here, we show that the nuclear receptor CAR can recognize response
Isabelle Dussault et al.
Molecular and cellular biology, 22(15), 5270-5280 (2002-07-09)
Unlike classical nuclear receptors that require ligand for transcriptional activity, the constitutive androstane receptor (CAR) is active in the absence of ligand. To determine the molecular contacts that underlie this constitutive activity, we created a three-dimensional model of CAR and
Jorge Luiz Vieira Dos Anjos et al.
International journal of pharmaceutics, 345(1-2), 81-87 (2007-06-30)
The interaction of a potent percutaneous penetration enhancer, 1,8-cineole, with the stratum corneum (SC) and DPPC membranes was investigated by electron paramagnetic resonance spectroscopy (EPR) of spin-labeled analogs of stearic acid (5-DSA) and androstanol (ASL). The EPR spectra of lipid
Andrea Toell et al.
Journal of cellular biochemistry, 85(1), 72-82 (2002-03-14)
Constitutive androstane receptor (CAR) and pregnane X receptor (PXR) are members of the nuclear receptor superfamily that regulate target gene transcription in a ligand-dependent manner. CAR and PXR have a rather broad, overlapping set of ligands that range from natural
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门


![氰(CN)离子标准液 traceable to SRM from NIST K₂[Zn(CN)₄] in H₂O 1000 mg/l CN Certipur®](/deepweb/assets/sigmaaldrich/product/images/920/032/af45eec3-100b-4996-8eb3-c3942d441bc9/640/af45eec3-100b-4996-8eb3-c3942d441bc9.jpg)