推荐产品
等級
primary reference standard
儲存期限
limited shelf life, expiry date on the label
製造商/商標名
HWI
應用
food and beverages
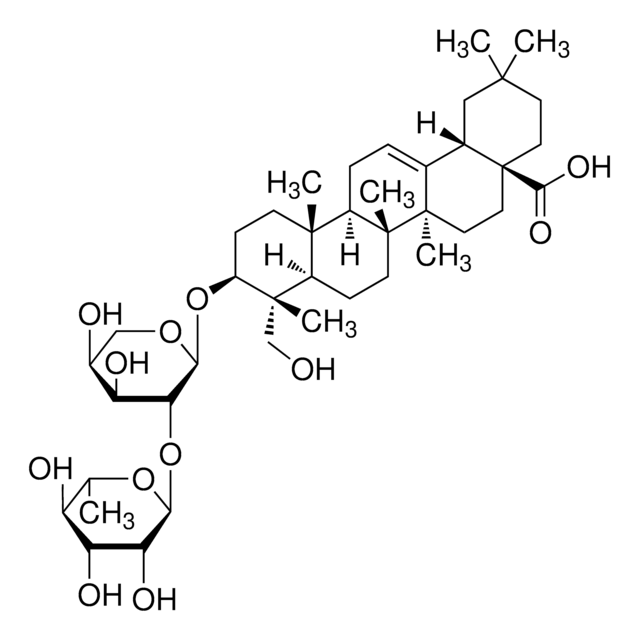
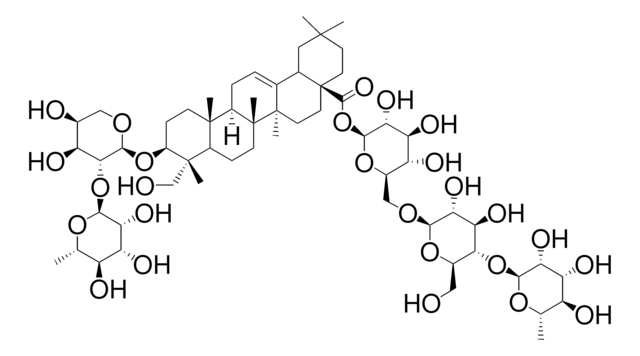
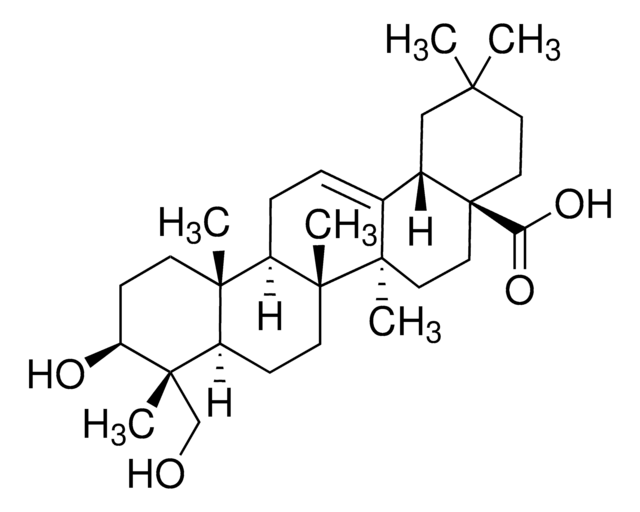
SMILES 字串
C[C@@H]1O[C@@H](O[C@@H]2[C@@H](O)[C@@H](O)CO[C@H]2O[C@H]3CC[C@@]4(C)[C@@H](CC[C@]5(C)[C@@H]4CC=C6[C@@H]7CC(C)(C)CC[C@@]7(CC[C@@]56C)C(O)=O)[C@]3(C)CO)[C@H](O)[C@H](O)[C@H]1O
InChI
1S/C41H66O12/c1-21-28(44)30(46)31(47)33(51-21)53-32-29(45)24(43)19-50-34(32)52-27-11-12-37(4)25(38(27,5)20-42)10-13-40(7)26(37)9-8-22-23-18-36(2,3)14-16-41(23,35(48)49)17-15-39(22,40)6/h8,21,23-34,42-47H,9-20H2,1-7H3,(H,48,49)/t21-,23-,24-,25+,26+,27-,28-,29-,30+,31+,32+,33-,34-,37-,38-,39+,40+,41-/m0/s1
InChI 密鑰
KEOITPILCOILGM-LLJOFIFVSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Exact content by quantitative NMR can be found on the certificate.
應用
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Oral
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
从最新的版本中选择一种:
分析证书(COA)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门