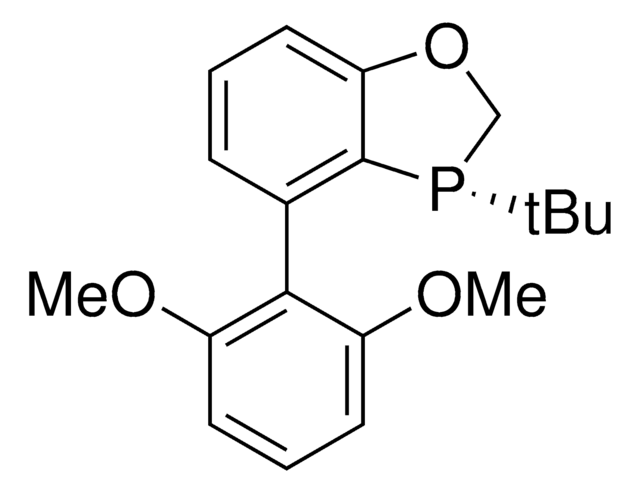
推荐产品
一般說明
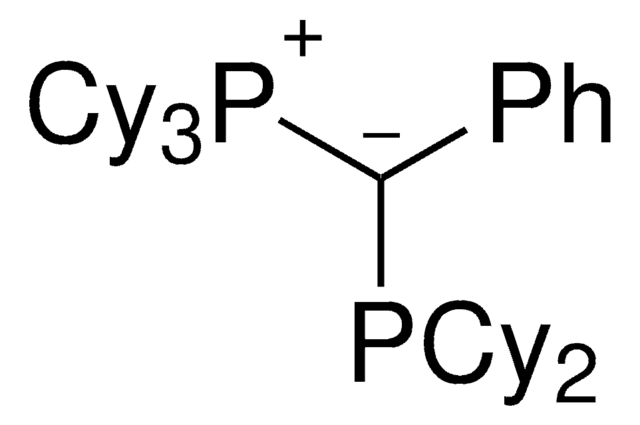
trYPhos™ is an ylide-functionalized phosphine ligand developed in the lab of Prof. V. Gessner with demonstrated uses in Pd-catalyzed cross coupling reactions, including the arylation of ketones and arylation of amines. trYPhos™is the strongest donor of the available YPhos™ ligands, and is suited towards the most challenging alpha-arylation reactions.
應用
The electron-rich and sterically demanding trYPhos™ ligand has been used in the gold(I)-catalyzed hydroamination of acetylene, and has shown to be effective in a range of Buchwald-Hartwig amination reactions. The strong electron-donor strength and sterically demanding nature of the ligand has been shown to increase the rate of formation of the catalytically active mono-phosphine palladium species, often leading to decreased reaction times or allowing the use of lower reaction temperatures. trYPhos™ is the most strongest donor of the available YPhos™ligands and has been found to be effective alpha-arylation of aliphatic cyclic ketones.
Learn more about ylide-functionalized phosphines (YPhos)
Learn more about ylide-functionalized phosphines (YPhos)
特點和優勢
Advantages of the trYPhos™ ligand over less electron rich ligand sources include, increased substrate scope in Buchwald-Hartwig amination reactions, including aryl chlorides, the use of more mild reaction conditions and improved activity in in C-N and C-C cross coupling reactions. The increased electron donor stregth of trYPhos™ has been shown to be effective in the challenging alpha-arylation of aliphatic ketones.
法律資訊
Product of Umicore
This product, its manufacturing or use, is the subject of one or more issued or pending U.S. Patents (and foreign equivalents) owned or controlled by Umicore PMC. The purchase of this product from Umicore PMC through Sigma-Aldrich, its affiliates or their authorized distributors conveys to the buyer a limited, one-time, non-exclusive, non-transferable, non-assignable license. Buyer′s use of this product may infringe patents owned or controlled by third parties. It is the sole responsibility of buyer to ensure that its use of the product does not infringe the patent rights of third parties or exceed the scope of the license granted herein.
For any further information on product please refer to your local Umicore PMC contact at www.pmc.umicore.com.
This product, its manufacturing or use, is the subject of one or more issued or pending U.S. Patents (and foreign equivalents) owned or controlled by Umicore PMC. The purchase of this product from Umicore PMC through Sigma-Aldrich, its affiliates or their authorized distributors conveys to the buyer a limited, one-time, non-exclusive, non-transferable, non-assignable license. Buyer′s use of this product may infringe patents owned or controlled by third parties. It is the sole responsibility of buyer to ensure that its use of the product does not infringe the patent rights of third parties or exceed the scope of the license granted herein.
For any further information on product please refer to your local Umicore PMC contact at www.pmc.umicore.com.
Yphos is a trademark of Umicore AG & Co. KG
相關產品
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Jens Tappen et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 26(19), 4281-4288 (2020-01-24)
Palladium allyl, cinnamyl, and indenyl complexes with the ylide-substituted phosphines Cy3 P+ -C- (R)PCy2 (with R=Me (L1) or Ph (L2)) and Cy3 P+ -C- (Me)PtBu2 (L3) were prepared and applied as defined precatalysts in C-N coupling reactions. The complexes are
Xiao-Qiang Hu et al.
Organic letters, 21(18), 7558-7562 (2019-08-31)
Ylide-functionalized phosphine (YPhos) ligands allow the palladium-catalyzed α-arylation of alkyl ketones with aryl chlorides with record setting activity. Using a cyclohexyl-substituted YPhos ligand, a wide range of challenging ketone substrates was efficiently and selectively monoarylated under mild conditions. A newly
Ilja Rodstein et al.
The Journal of organic chemistry, 85(22), 14674-14683 (2020-09-11)
Ylide-substituted phosphines have been shown to be excellent ligands for C-N coupling reactions under mild reaction conditions. Here we report studies on the impact of the steric demand of the substituent in the ylide-backbone on the catalytic activity. Two new
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门
![[Pd(IPr#)(cin)Cl]](/deepweb/assets/sigmaaldrich/product/structures/391/578/9bb7eaef-fa70-4f50-8644-2c55cec3925d/640/9bb7eaef-fa70-4f50-8644-2c55cec3925d.png)