P7768
Pyruvate Kinase from rabbit muscle
Type VII, buffered aqueous glycerol solution, 350-600 units/mg protein
Synonyme(s) :
ATP:pyruvate 2-O-phosphotransferase, PK
About This Item
Produits recommandés
Type
Type VII
Niveau de qualité
Forme
buffered aqueous glycerol solution
Activité spécifique
350-600 units/mg protein
Poids mol.
237 kDa
Concentration
2.0-20.0 mg/mL
Activité étrangère
lactic dehydrogenase and creatine phosphokinase ≤0.01%
phosphoglucomutase and myokinase ≤0.05%
Température de stockage
2-8°C
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
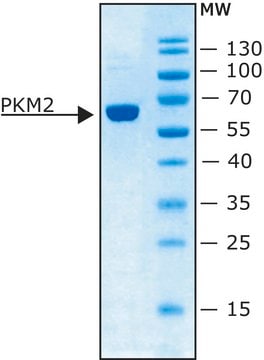
Pyruvate kinase plays a role in regulating cell metabolism. There are four pyruvate kinase isoforms in mammals (PKM1, PKM2, PKR, PKL). Mammalian pyruvate kinase is a tetrameric protein composed of identical subunits, arranged in a dimer-of-dimers configuration. Each monomer contains one active site and consists of three main domains- designated A, B, and C-along with a small N-terminal domain. The M2 isoform of pyruvate kinase (PKM2) supports anabolic metabolism and is expressed in cancer and normal tissue.
Application
Actions biochimiques/physiologiques
Isoelectric Point: 7.6
Optimal pH: ∼7.5
Optimal Temperature: 25°C
ΕA280 = 0.54 for 1 mg(p)/ml, 1 cm path
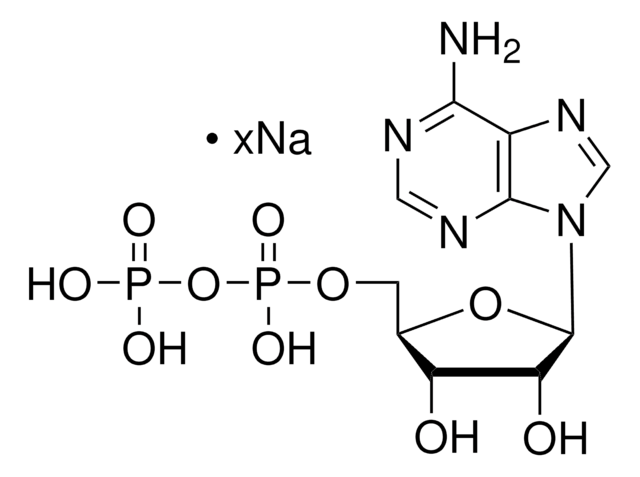
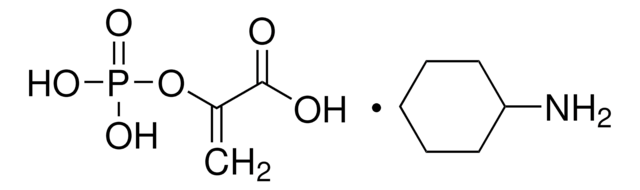
Reported KM values are ATP (0.86 mM), pyruvate (10 mM), ADP (0.3 mM), and PEP (0.07 mM) in Tris buffer at pH 7.4 and 30 °C. Pyruvate kinase is highly specific for phosphoenolpyruvate, but can utilize other dinucleotide triphosphates as substrates in place of ATP including GTP, ITP, dATP, UTP, and CTP.
Isoelectric Point: 7.6
Optimal pH: ∼7.5
Définition de l'unité
Forme physique
Remarque sur l'analyse
Code de la classe de stockage
10 - Combustible liquids
Classe de danger pour l'eau (WGK)
WGK 2
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique