196487
4-Biphenylacetic acid
98%
About This Item
Produits recommandés
Niveau de qualité
Essai
98%
Pf
159-160 °C (lit.)
Solubilité
DMSO: soluble 50 mg/mL, clear, colorless to yellow
Chaîne SMILES
OC(=O)Cc1ccc(cc1)-c2ccccc2
InChI
1S/C14H12O2/c15-14(16)10-11-6-8-13(9-7-11)12-4-2-1-3-5-12/h1-9H,10H2,(H,15,16)
Clé InChI
QRZAKQDHEVVFRX-UHFFFAOYSA-N
Informations sur le gène
human ... BAD(572)
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique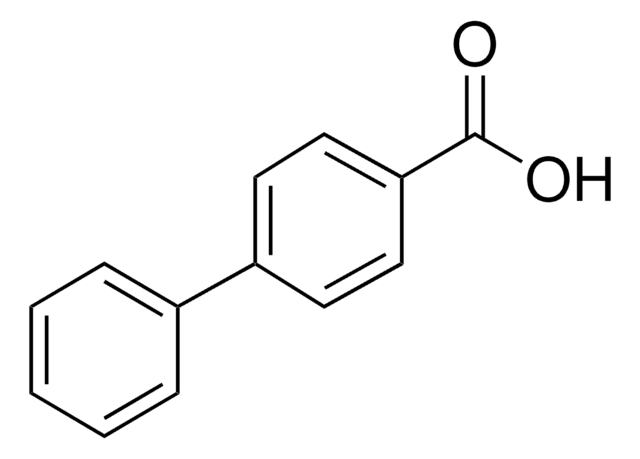


![2-[2-(2-Chloroethoxy)ethoxy]ethanol 96%](/deepweb/assets/sigmaaldrich/product/structures/902/295/ff6d7bb1-a7e0-4582-86e1-819084626e67/640/ff6d7bb1-a7e0-4582-86e1-819084626e67.png)

