767344
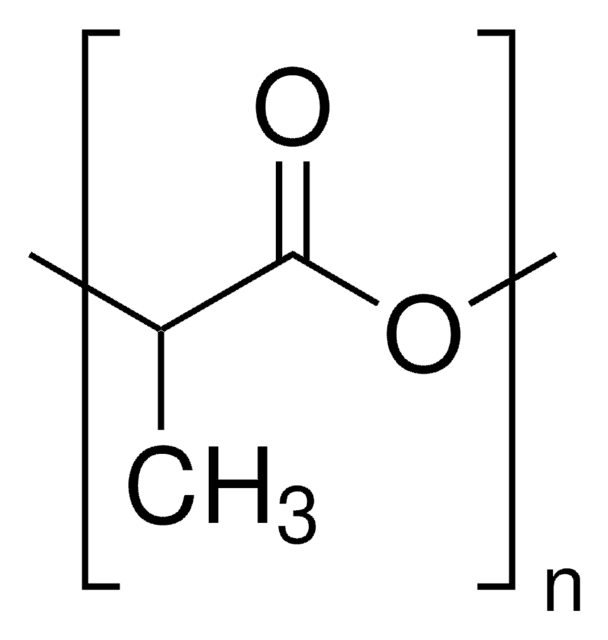
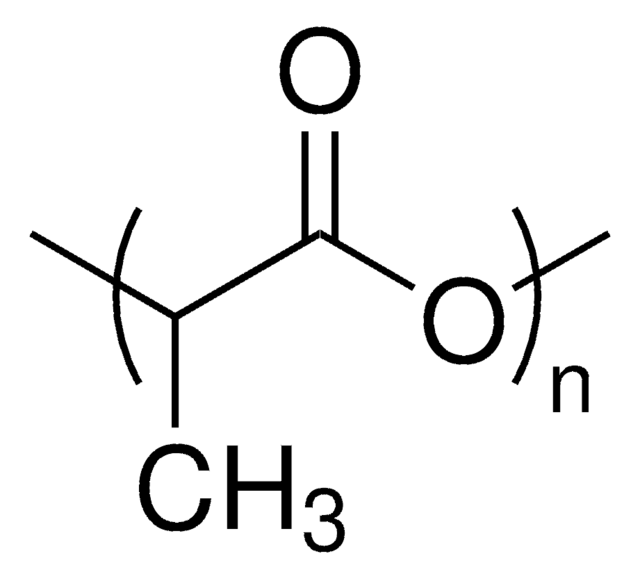
Poly(D,L-lactide)
average Mn 20,000 (NMR), PDI ≤1.3
Synonym(s):
PDLA, PLA, polyDL-lactide
About This Item
Recommended Products
form
crystals
Quality Level
mol wt
average Mn 20,000 (NMR)
degradation timeframe
<6 months
mp
320-350 °C
PDI
≤1.3
storage temp.
2-8°C
Looking for similar products? Visit Product Comparison Guide
Application
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Local delivery of bioactive molecules using an implantable device can decrease the amount of drug dose required as well as non-target site toxicities compared to oral or systemic drug administration.
Microparticle drug delivery systems have been extensively researched and applied to a wide variety of pharmaceutical and medical applications due to a number of advantages including injectability, local applicability to target tissues and sites, and controlled drug delivery over a given time period.
The world of commercial biomaterials has stagnated over the past 30 years as few materials have successfully transitioned from the bench to clinical use. Synthetic aliphatic polyesters have continued to dominate the field of resorbable biomaterials due to their long history and track record of approval with the U.S. Food and Drug Administration (FDA).
Aliphatic polyesters such as polylactide, poly(lactide-co-glycolide) and polycaprolactone, as well as their copolymers, represent a diverse family of synthetic biodegradable polymers that have been widely explored for medical uses and are commercially available.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service