SML1779
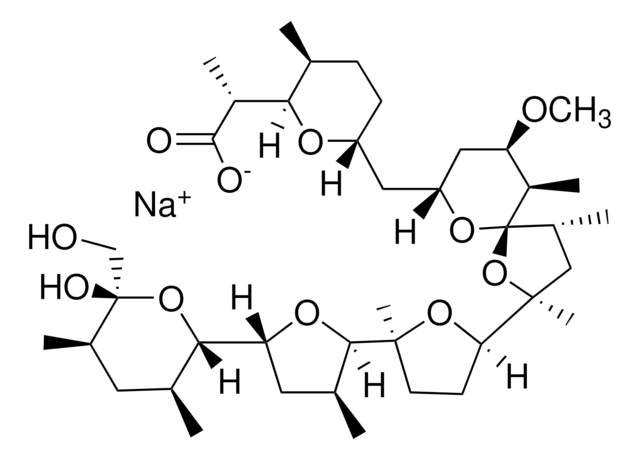
Nigericin sodium salt
from Streptomyces hygroscopicus, ≥98% (HPLC), solution, polyether ionophore
Sinónimos:
3B2-6379, Antibiotic K178, Antibiotic X464, Azalomycin M, HE331800, Helexin C, Polyetherin A, sodium;2-[(3S,6R)-6-[[(5R,6R,7R,9R)-2-[5-[(3S,5R)-5-[(2S,3S,5R,6R)-6-hydroxy-6-(hydroxymethyl)-3,5-dimethyloxan-2-yl]-3-methyloxolan-2-yl]-5-methyloxolan-2-yl]-7-methoxy-2,4,6-trimethyl-1,10-dioxaspiro[4.5]decan-9-yl]methyl]-3-methyloxan-2-yl]propanoate
About This Item
Productos recomendados
product name
Nigericin sodium salt Ready Made Solution, 5 mg/mL (DMSO:ethanol 1:1)
origen biológico
Streptomyces hygroscopicus
Nivel de calidad
formulario
solution
concentración
5 mg/mL (DMSO:ethanol 1:1)
espectro de actividad antibiótica
Gram-positive bacteria
Modo de acción
cell membrane | interferes
Condiciones de envío
ambient
temp. de almacenamiento
2-8°C
InChI
1S/C40H68O11.Na/c1-21-11-12-28(46-33(21)26(6)36(42)43)17-29-18-30(45-10)27(7)40(48-29)25(5)19-38(9,51-40)32-13-14-37(8,49-32)35-23(3)16-31(47-35)34-22(2)15-24(4)39(44,20-41)50-34;/h21-35,41,44H,11-20H2,1-10H3,(H,42,43);/q;+1/p-1/t21-,22-,23-,24+,25?,26?,27+,28+,29+,30+,31+,32?,33?,34-,35?,37?,38?,39-,40+;/m0./s1
Clave InChI
MOYOTUKECQMGHE-KKCUGXASSA-M
Categorías relacionadas
Acciones bioquímicas o fisiológicas
Nigericin kills bacteria by facilitating the diffusion of ions across membranes.
Low concentration (0.5 μM) of Nigericin rapidly decreases pHi, causing stimulation of PG production 1.5- to 2-fold in cerebral microvascular endothelial cells and arresting of DNA synthesis in Erlich acites carcinoma cells. Treatment of Hela cells, after entry of poliovirus, with nigericin, prevents the inhibition of host protein synthesis by poliovirus. Nigericin is also widely used in studies of the consequences of changes in membrane potential in variable systems.
Nota de preparación
Almacenamiento y estabilidad
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Flam. Liq. 3
Código de clase de almacenamiento
3 - Flammable liquids
Clase de riesgo para el agua (WGK)
WGK 2
Punto de inflamabilidad (°F)
78.8 °F
Punto de inflamabilidad (°C)
26 °C
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico