400091
DMOG
≥98% (HPLC), solid, HIF-hydroxylase inhibitor, Calbiochem
Sinónimos:
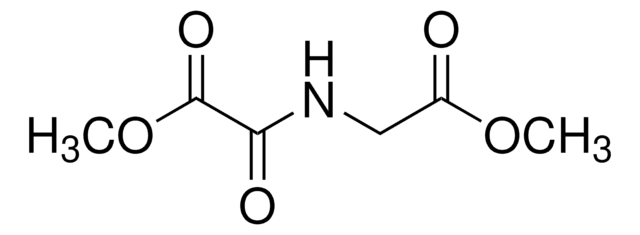
HIF-Hydroxylase Inhibitor, DMOG, N-(Methoxyoxoacetyl)-glycine methyl ester, Dimethyloxalylglycine, HIF Prolyl Hydroxylase Inhibitor I, HIF Aspartyl β-Hydroxylase Inhibitor, HIF Asparanginyl β-Hydroxylase Inhibitor
About This Item
Productos recomendados
Nombre del producto
HIF-Hydroxylase Inhibitor, DMOG, The HIF-Hydroxylase Inhibitor, DMOG, also referenced under CAS 89464-63-1, controls the biological activity of HIF-Hydroxylase. This small molecule/inhibitor is primarily used for Cell Structure applications.
Nivel de calidad
Ensayo
≥98% (HPLC)
fabricante / nombre comercial
Calbiochem®
condiciones de almacenamiento
OK to freeze
desiccated (hygroscopic)
color
pink-white to peach-white
solubilidad
DMSO: 50 mg/mL
Condiciones de envío
ambient
temp. de almacenamiento
2-8°C
InChI
1S/C6H9NO5/c1-11-4(8)3-7-5(9)6(10)12-2/h3H2,1-2H3,(H,7,9)
Clave InChI
BNJOZDZCRHCODO-UHFFFAOYSA-N
Descripción general
Advertencia
Reconstitución
Otras notas
Chen, H., et al. 2006. Cancer Res.66, 9009.
Milkiewicz, M., et al. 2004. J. Physiol.560, 21.
Lando, D., et al. 2002. Science295, 858.
Jaakkola, P., et al. 2001. Science292, 468.
Información legal
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico