595314
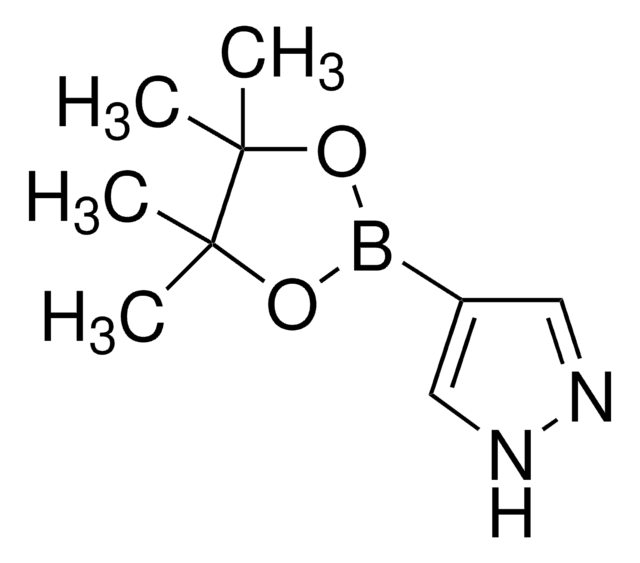
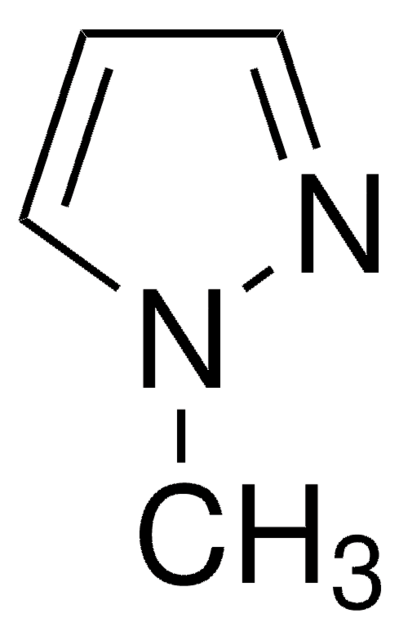
1-Methylpyrazole-4-boronic acid pinacol ester
95%
Sinónimos:
1-Methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 1-Methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole, 1-Methyl-4-pyrazoleboronic acid pinacol ester, 2-(1-Methylpyrazol-4-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane, 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1-methylpyrazole
About This Item
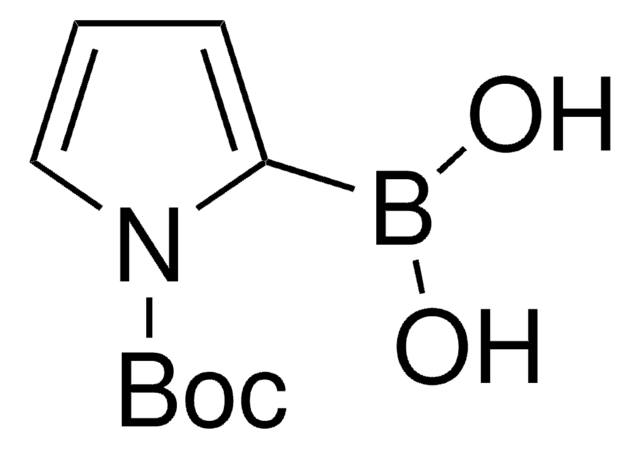
Productos recomendados
Nivel de calidad
Ensayo
95%
Formulario
solid
mp
59-64 °C (lit.)
cadena SMILES
Cn1cc(cn1)B2OC(C)(C)C(C)(C)O2
InChI
1S/C10H17BN2O2/c1-9(2)10(3,4)15-11(14-9)8-6-12-13(5)7-8/h6-7H,1-5H3
Clave InChI
UCNGGGYMLHAMJG-UHFFFAOYSA-N
Categorías relacionadas
Aplicación
- Suzuki-Miyaura cross-coupling reactions
- Transesterification reactions
Reagent used for preparation of
- Aminothiazoles as γ-secretase modulators
- Amino-pyrido-indol-carboxamides, as potential JAK2 inhibitors for myeloproliferative disorders therapy
- Pyridine derivatives as TGF-β1 and activin A signalling inhibitors
- MK-2461 analogs as inhibitors of c-Met kinase for the treatment of cancer
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
This brochure contains a comprehensive selection of boronic acids, boronic acid esters, diboron esters, and transition-metal catalysts useful for the Suzuki–Miyaura coupling reaction
The Suzuki-Miyaura cross-coupling reaction is an important and extensively used reaction in organic chemistry with applications in polymer science and in the fine chemicals and pharmaceutical industries.
The synthesis of biaryl compounds via the Suzuki–Miyaura coupling reaction has become more commonplace now that many arylboronic acids are readily available.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico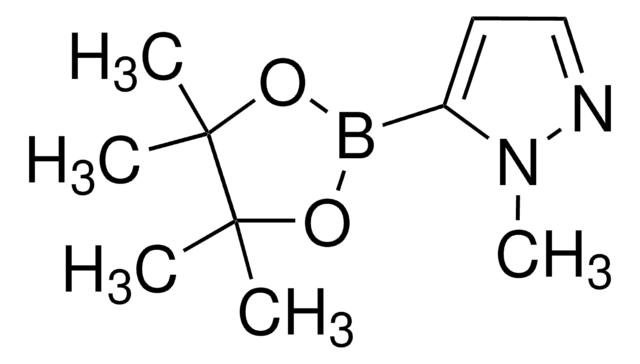
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

![1-[1-(2-Methylphenyl)-1H-pyrazol-4-yl]methanamine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/193/071/8c363ad6-8306-4c4d-b322-749ff2feff6f/640/8c363ad6-8306-4c4d-b322-749ff2feff6f.png)
![4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrrolo[2,3-b]pyridine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/272/176/ea333f93-763d-458c-a328-3969b7d46e5d/640/ea333f93-763d-458c-a328-3969b7d46e5d.png)