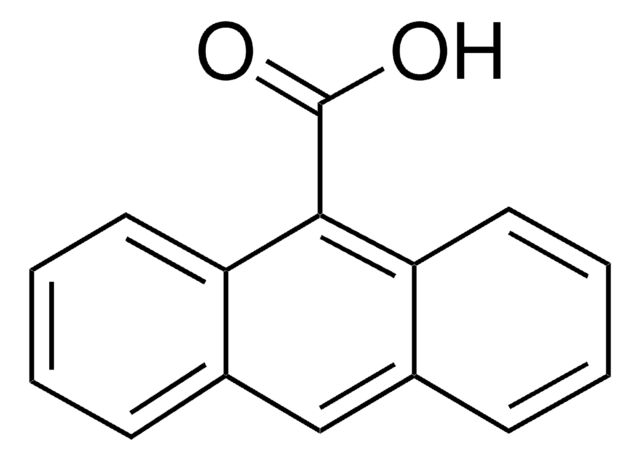
392189
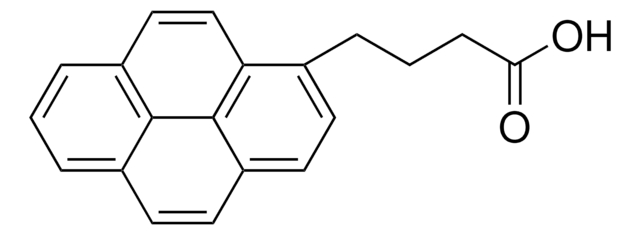
1-Pyreneacetic acid
97%
Sinónimos:
(1-Pyrenyl)acetic acid
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C18H12O2
Número de CAS:
Peso molecular:
260.29
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
solid
mp
210-212 °C (dec.) (lit.)
grupo funcional
carboxylic acid
cadena SMILES
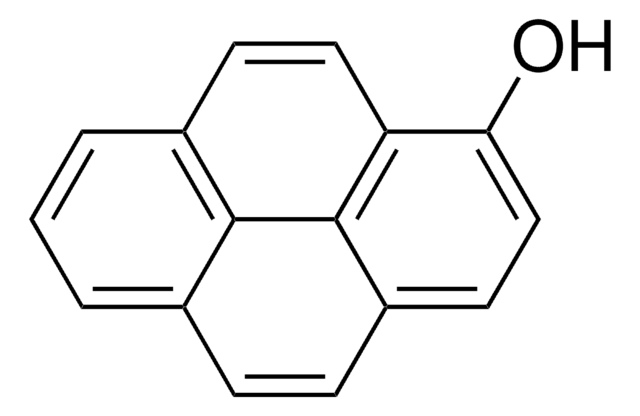
OC(=O)Cc1ccc2ccc3cccc4ccc1c2c34
InChI
1S/C18H12O2/c19-16(20)10-14-7-6-13-5-4-11-2-1-3-12-8-9-15(14)18(13)17(11)12/h1-9H,10H2,(H,19,20)
Clave InChI
SDJCLYBBPUHKCD-UHFFFAOYSA-N
Categorías relacionadas
Descripción general
1-Pyreneacetic acid is a negatively charged pyrene derivative. It has been proposed as titrating reagent for the standardization titration of Grignard reagents and n-butyl lithium (n-BuLi).
Aplicación
1-Pyreneacetic acid is suitable for use in the following studies:
- Synthesis of N-(2-(methylthio)ethyl)-2-(pyren-1-yl)acetamide, a pyrene amide based Pd2+ probe.
- Synthesis of pyrene-modified β-cyclodextrin.
- To functionalize single walled carbon nanotube field effect transistors (CNT FETs).
- As an agent for characterizing grafting degrees and reactivity of the ester functionalized polypropylenes.
- Synthesis sawhorse-type diruthenium tetracarbonyl complexes.
- Synthesis of (±)-2-(1-pyrenyl)propionic acid, a chiral carboxylic acid.
- Reversible noncovalent functionalization of single walled carbon nanotubes (SWNTs).
- Preparation of peptide nucleic acid (PNA) probes.
- As an internal reference compound in the assessment of solid phase reaction by HPLC-UV.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Jan Spengler et al.
ACS combinatorial science, 15(5), 229-234 (2013-03-26)
Here we evaluated the use of internal reference compounds for the rapid assessment of reactions performed in solid-phase. An internal reference compound (commercially available) was bound to the resin, together with the substrate, and cleaved with the products after completion
Sawhorse-type diruthenium tetracarbonyl complexes derived from pyrenyl-carboxylic acids.
Johnpeter JP and Therrien B.
Inorgorganica Chimica Acta, 405, 437-443 (2013)
Gabriela Ramos Chagas et al.
Chemphyschem : a European journal of chemical physics and physical chemistry, 18(23), 3429-3436 (2017-09-01)
A smart stimuli-responsive surface was fabricated by the electro-copolymerization of pyrene monomers followed by base and acid treatment. Copolymers of pyrenes bearing fluorinated chains (Py-nF
Alex Manicardi et al.
Beilstein journal of organic chemistry, 10, 1495-1503 (2014-08-28)
Pyrene derivatives can be incorporated into nucleic acid analogs in order to obtain switchable probes or supramolecular architectures. In this paper, peptide nucleic acids (PNAs) containing 1 to 3 1-pyreneacetic acid units (PNA1-6) with a sequence with prevalence of pyrimidine
Murphy PJ.
Organophosphorus Reagents: A Practical Approach in Chemistry, 8-8 (2004)
Global Trade Item Number
| Número de referencia del producto (SKU) | GTIN |
|---|---|
| 392189-1G | 4061825923079 |
| 392189-5G | 4061832619149 |
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico