375217
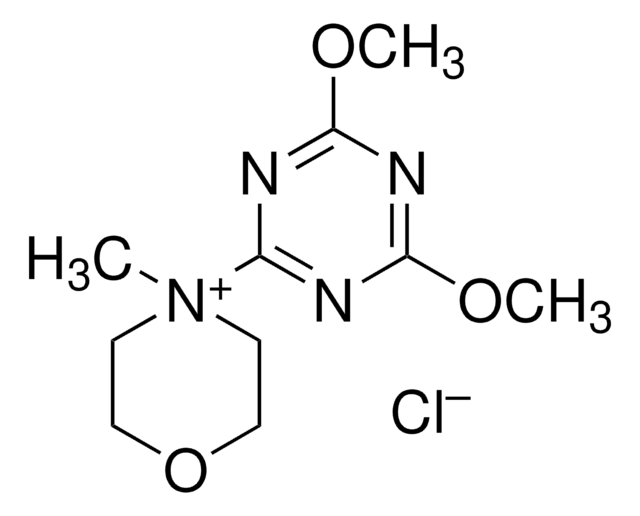
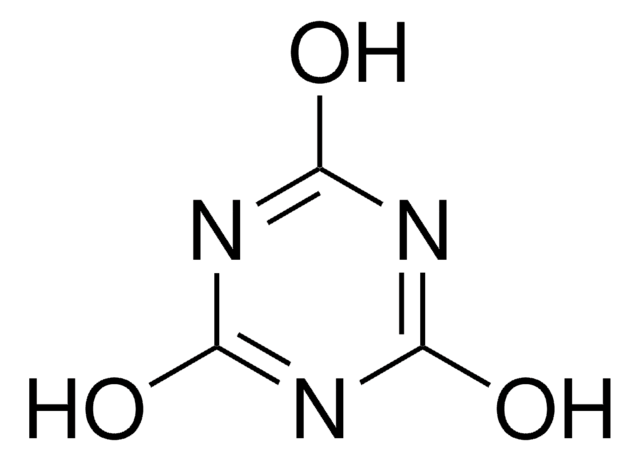
2-Chloro-4,6-dimethoxy-1,3,5-triazine
97%
Sinónimos:
2,4-Dimethoxy-6-chloro-1,3,5-triazine, 2,4-Dimethoxy-6-chloro-s-triazine, 2-Chloro-4,6-dimethoxy-1,3,5-triazine, 2-Chloro-4,6-dimethoxy-s-triazine, 4,6-Dimethoxy-2-chloro-s-triazine, 6-Chloro-2,4-dimethoxy-s-triazine, CDMT, Chlorodimethoxytriazine
About This Item
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
solid
mp
71-74 °C (lit.)
grupo funcional
chloro
cadena SMILES
COc1nc(Cl)nc(OC)n1
InChI
1S/C5H6ClN3O2/c1-10-4-7-3(6)8-5(9-4)11-2/h1-2H3
Clave InChI
GPIQOFWTZXXOOV-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
- The specific labeling of streptavidin by the modular method for affinity labeling (MoAL).
- The preparation of bis(4,6-dimethoxy-1,3,5-triazin-2-yl) ether, via reaction with 2-hydroxy-4,6-dimethoxy-1,3,5-triazine.
- The preparation of 2-(4,6-dimethoxy-1,3,5-triazinyl)trialkylammonium salts, via reaction with various tertiary amines.
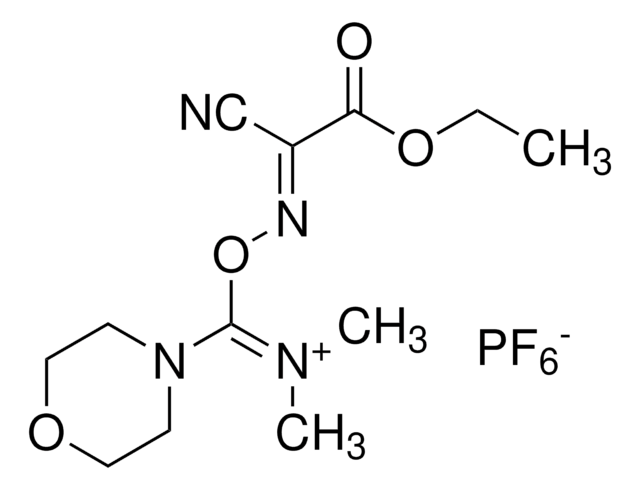
- The synthesis of 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride, via coupling with N-methylmorpholine in THF.
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1
Código de clase de almacenamiento
8A - Combustible corrosive hazardous materials
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Global Trade Item Number
| Número de referencia del producto (SKU) | GTIN |
|---|---|
| 375217-25G | |
| 375217-1G | |
| 375217-5G | 4061831834963 |
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico