250031
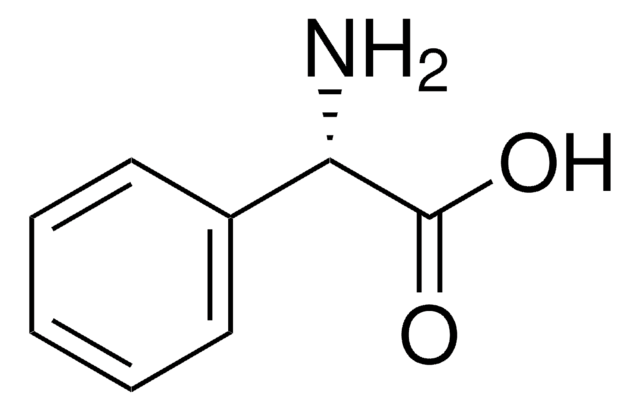
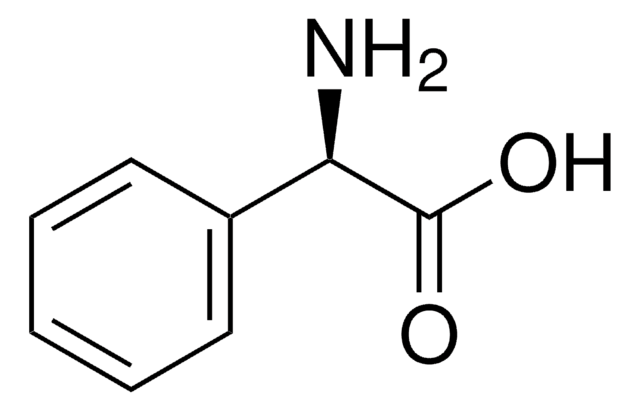
(R)-(−)-N-(3,5-Dinitrobenzoyl)-α-phenylglycine
99%, for peptide synthesis
Sinónimos:
N-(3,5-Dinitrobenzoyl)-D-α-phenylglycine, R-(−)-N-(3,5-Dinitrobenzoyl)phenylglycine
About This Item
Productos recomendados
Nombre del producto
(R)-(−)-N-(3,5-Dinitrobenzoyl)-α-phenylglycine, 99%
Ensayo
99%
actividad óptica
[α]20/D −102°, c = 0.9 in THF
idoneidad de la reacción
reaction type: solution phase peptide synthesis
mp
216-217 °C (lit.)
aplicaciones
peptide synthesis
cadena SMILES
OC(=O)[C@H](NC(=O)c1cc(cc(c1)[N+]([O-])=O)[N+]([O-])=O)c2ccccc2
InChI
1S/C15H11N3O7/c19-14(16-13(15(20)21)9-4-2-1-3-5-9)10-6-11(17(22)23)8-12(7-10)18(24)25/h1-8,13H,(H,16,19)(H,20,21)/t13-/m1/s1
Clave InChI
MIVUDAUOXJDARR-CYBMUJFWSA-N
Categorías relacionadas
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico