Wichtige Dokumente
U0383
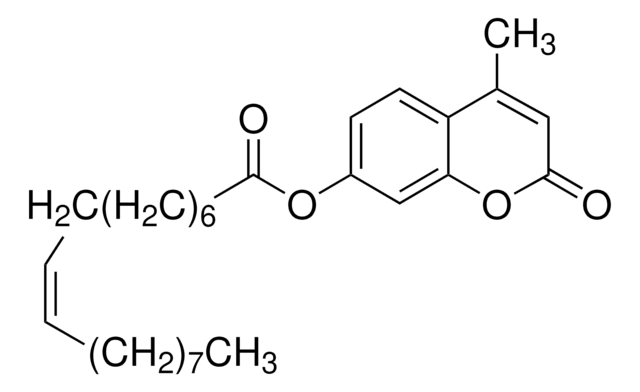
Umbelliferyl Arachidonate
ethanol solution
Synonym(e):
5Z,8Z,11Z,14Z-Eicosatetraenoic acid 2-oxo-2H-1-benzopyran-7-yl ester, 7-Hydroxycoumarinyl Arachidonate
About This Item
Empfohlene Produkte
Biologische Quelle
synthetic (organic)
Qualitätsniveau
Assay
≥98% (HPLC)
Form
ethanol solution
Methode(n)
immunofluorescence: suitable
Löslichkeit
DMSO: soluble 25 mg/mL
DMF: soluble 50 mg/mL
aqueous buffer: slightly soluble
Versandbedingung
wet ice
Lagertemp.
−20°C
SMILES String
CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(=O)Oc1ccc2C=CC(=O)Oc2c1
InChI
1S/C29H36O4/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-28(30)32-26-22-20-25-21-23-29(31)33-27(25)24-26/h6-7,9-10,12-13,15-16,20-24H,2-5,8,11,14,17-19H2,1H3/b7-6-,10-9-,13-12-,16-15-
InChIKey
SFTGFGOBCQCZDY-DOFZRALJSA-N
Anwendung
Biochem./physiol. Wirkung
Angaben zur Herstellung
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Flam. Liq. 2
Lagerklassenschlüssel
3 - Flammable liquids
WGK
WGK 2
Flammpunkt (°F)
57.2 °F - closed cup
Flammpunkt (°C)
14 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.