Wichtige Dokumente
860493P
Avanti
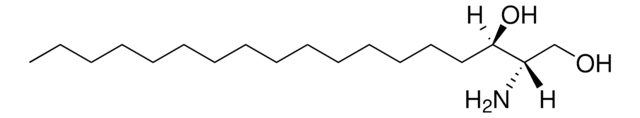
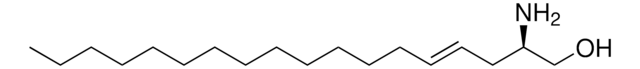
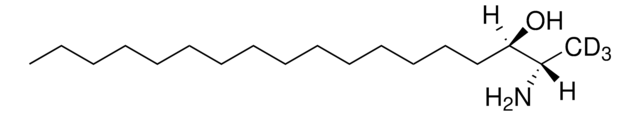
1-deoxysphinganine
Avanti Research™ - A Croda Brand 860493P, powder
Synonym(e):
1-deoxysphinganine (m18:0)
About This Item
Empfohlene Produkte
Form
powder
Verpackung
pkg of 1 × 1 mg (860493P-1mg)
pkg of 1 × 10 mg (860493P-10mg)
Hersteller/Markenname
Avanti Research™ - A Croda Brand 860493P
Lipid-Typ
bioactive lipids
sphingolipids
Versandbedingung
dry ice
Lagertemp.
−20°C
SMILES String
C[C@@](N)([H])[C@]([H])(O)CCCCCCCCCCCCCCC
InChI
1S/C18H39NO/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-18(20)17(2)19/h17-18,20H,3-16,19H2,1-2H3/t17-,18+/m0/s1
InChIKey
YRYJJIXWWQLGGV-ZWKOTPCHSA-N
Allgemeine Beschreibung
Anwendung
Biochem./physiol. Wirkung
Verpackung
Rechtliche Hinweise
Lagerklassenschlüssel
11 - Combustible Solids
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Leider sind derzeit keine COAs für dieses Produkt online verfügbar.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.