Alle Fotos(3)
Wichtige Dokumente
O9808
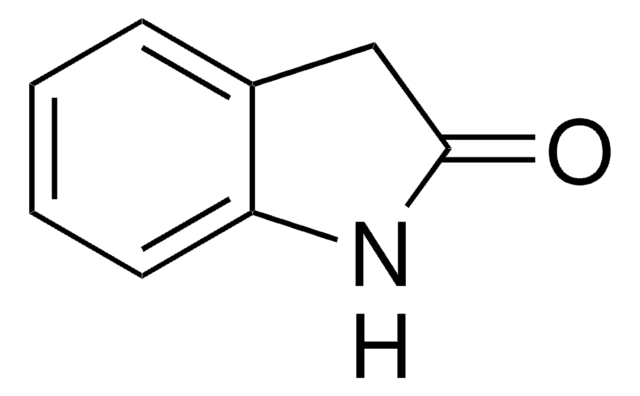
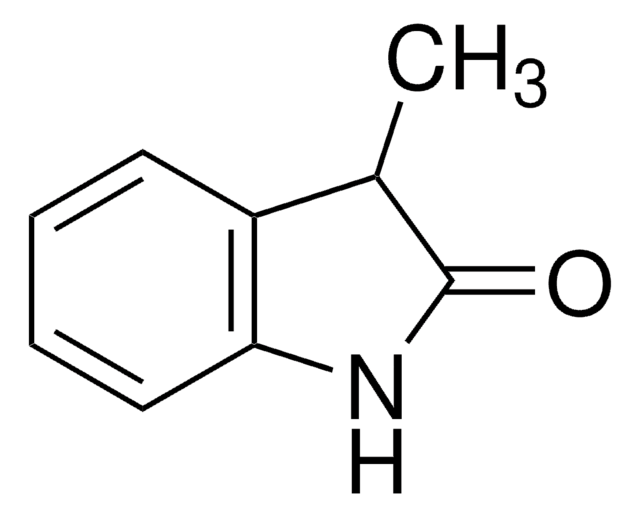
2-Indolinon
97%
Synonym(e):
2-Oxindol, Oxindol
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(3)
About This Item
Empirische Formel (Hill-System):
C8H7NO
CAS-Nummer:
Molekulargewicht:
133.15
Beilstein:
114692
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
97%
Form
crystals
bp
227 °C/73 mmHg (lit.)
mp (Schmelzpunkt)
123-128 °C (lit.)
SMILES String
O=C1Cc2ccccc2N1
InChI
1S/C8H7NO/c10-8-5-6-3-1-2-4-7(6)9-8/h1-4H,5H2,(H,9,10)
InChIKey
JYGFTBXVXVMTGB-UHFFFAOYSA-N
Angaben zum Gen
human ... PGR(5241)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Junwei Wang et al.
Organic letters, 14(9), 2210-2213 (2012-04-14)
The reaction of phenyliodine bis(trifluoroacetate) (PIFA) with a series of anilides 1 (E = CO(2)Et) in CF(3)CH(2)OH was found to give 3-hydroxy-2-oxindole derivatives 2, while that with various anilides 1' (E = CON(R(4))Ar) afforded the C(2)-symmetric or unsymmetric spirooxindoles 3.
Ryo Shintani et al.
Chemical communications (Cambridge, England), 46(36), 6822-6824 (2010-08-21)
A copper-catalyzed addition of arylboronates to isatins has been developed to give 3-aryl-3-hydroxy-2-oxindoles under mild conditions. The catalytic cycle of this process has been examined through a series of stoichiometric reactions and an effective asymmetric variant has also been described
Rahul R Khanwelkar et al.
Bioorganic & medicinal chemistry, 18(13), 4674-4686 (2010-06-24)
A series of new ureidoindolin-2-one derivatives were synthesized and evaluated as inhibitors of receptor tyrosine kinases. Investigation of structure-activity relationships at positions 5, 6, and 7 of the oxindole skeleton led to the identification of 6-ureido-substituted 3-pyrrolemethylidene-2-oxindole derivatives that potently
Morteza Bararjanian et al.
The Journal of organic chemistry, 75(9), 2806-2812 (2010-04-15)
An efficient palladium-catalyzed protocol for the synthesis of 3-arylidene-2-oxindoles has been developed. In this approach, a sequential one-pot six-component reaction via Ugi/Heck carbocyclization/Sonogashira/nucleophilic addition was used for the synthesis of the desired skeleton.
C Lanzi et al.
International journal of cancer, 85(3), 384-390 (2000-02-01)
ret-derived oncogenes are frequently and specifically expressed in thyroid tumors. In contrast to the ret receptor, ret oncoproteins are characterized by ligand-independent tyrosine-kinase activity and tyrosine phosphorylation. In this study, novel synthetic arylidene 2-indolinone compounds were evaluated as inhibitors of
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.