424447
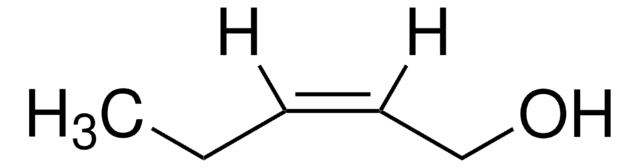
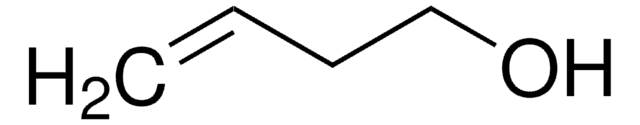
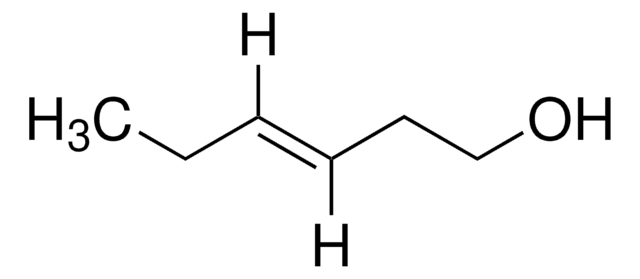
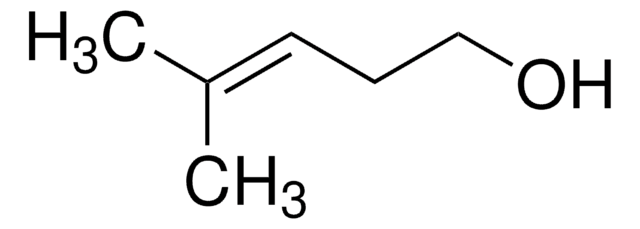
trans-2-Penten-1-ol
95%
Synonym(e):
(2E)-2-Penten-1-ol, (E)-Pent-2-en-1-ol, trans-2-Pentenol
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Lineare Formel:
C2H5CH=CHCH2OH
CAS-Nummer:
Molekulargewicht:
86.13
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
95%
Form
liquid
Brechungsindex
n20/D 1.434 (lit.)
bp
139-139.5 °C (lit.)
Dichte
0.847 g/mL at 25 °C (lit.)
Funktionelle Gruppe
hydroxyl
SMILES String
CC\C=C\CO
InChI
1S/C5H10O/c1-2-3-4-5-6/h3-4,6H,2,5H2,1H3/b4-3+
InChIKey
BTSIZIIPFNVMHF-ONEGZZNKSA-N
Allgemeine Beschreibung
trans-2-Penten-1-ol is an allyl alcohol. It is one of the volatile compounds found in olive oil, cashew apple juice and fermented cucumber brines. The rate constants and product ion distributions of its reaction with H3O+, NO+ and O2.+ ions have been studied using selected ion flow tube (SIFT).
Anwendung
trans-2-Penten-1-ol may be used in the synthesis of the following:
- leustroducsin B
- trichloroacetimidate
- (E)-2,3,3′-trifluoro-4-(2-(trans-4-pentylcyclohexyl)ethyl)-4′-(pent-2-enyloxy)biphenyl
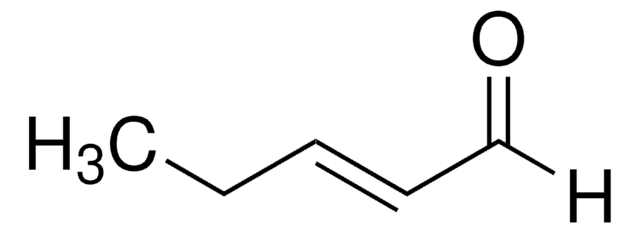
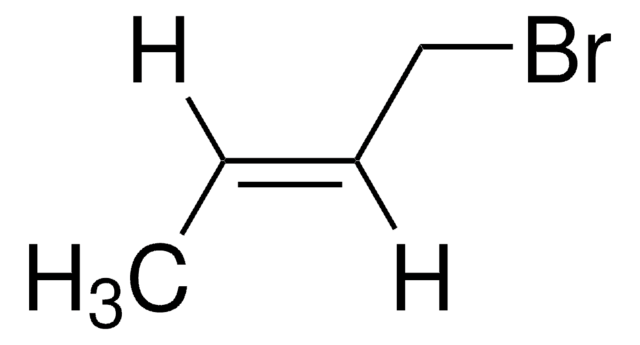
- trans-1-bromo-2-pentene
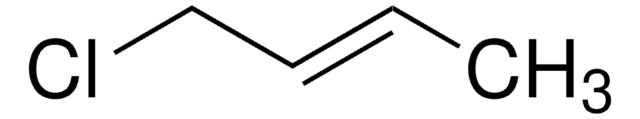
- trans-1-chloro-2-pentene
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Kazuyuki Miyashita et al.
The Journal of organic chemistry, 73(14), 5360-5370 (2008-06-14)
Leustroducsin B was synthesized via a convergent route based on division of the leustroducsin molecule into three segments A, B, and C. Two coupling reactions (Julia coupling reaction and Nozaki-Hiyama-Kishi (NHK) reaction) were employed for coupling of segments A and
Suzanne D Johanningsmeier et al.
Journal of food science, 76(1), C168-C177 (2011-05-04)
A nontargeted, comprehensive 2-dimensional gas chromatography-time-of-flight mass spectrometry (GC×GC-TOFMS) method was developed for the analysis of fermented cucumber volatiles before and after anaerobic spoilage. Volatile compounds extracted by solid-phase microextraction were separated on a polyethylene glycol 1st-dimension column and 14%
Biogeneration of volatile compounds in virgin olive oil: their evolution in relation to malaxation time.
Angerosa F, et al.
Journal of Agricultural and Food Chemistry, 46(8), 2940-2944 (1998)
A selected ion flow tube study of the reactions of H3O+, NO+ and O2.+ with a series of C5, C6 and C8 unsaturated biogenic alcohols.
Schoon N, et al.
International Journal of Mass Spectrometry, 263(2-3), 127-136 (2007)
Stereoregulated synthesis of unsaturated compounds Communication 9. Stereochemistry of the reactions of aldehydes with ?, ?-unsaturated triphenylphosphonium ylides [alkylidenetriphenylphosphoranes].
Bergel'son LD, et al.
Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 15(3), 468-473 (1966)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.