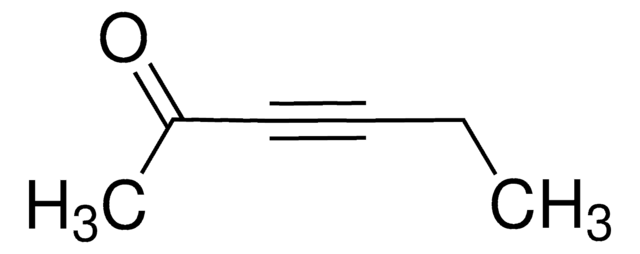Alle Fotos(1)
Wichtige Dokumente
418676
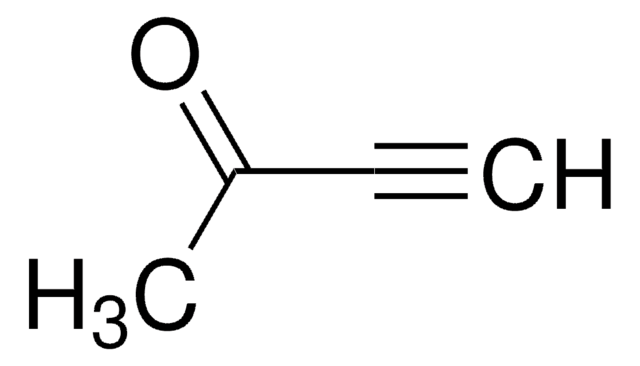
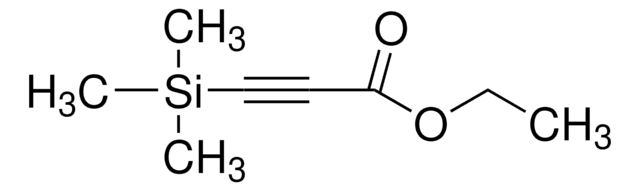
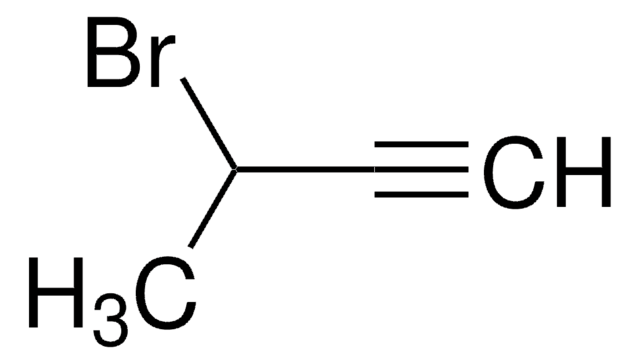
4-(Trimethylsilyl)-3-butin-2-on
97%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
(CH3)3SiC≡CCOCH3
CAS-Nummer:
Molekulargewicht:
140.26
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
97%
Form
liquid
Brechungsindex
n20/D 1.442 (lit.)
bp
156 °C (lit.)
Dichte
0.854 g/mL at 25 °C (lit.)
Funktionelle Gruppe
ketone
SMILES String
CC(=O)C#C[Si](C)(C)C
InChI
1S/C7H12OSi/c1-7(8)5-6-9(2,3)4/h1-4H3
InChIKey
NQEZDDPEJMKMOS-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
4-(Trimethylsilyl)-3-butyn-2-one is a ketone. Its asymmetric bioreduction to enantiopure {(S)-TMSBOL in various hydrophilic ionic liquid (ILs) solvent systems has been reported.
Anwendung
4-(Trimethylsilyl)-3-butyn-2-one (TMSB) has been used to investigate its asymmetric bioreduction to (S)-4-(trimethylsilyl)-3-butyn-2-ol {(S)-TMSBOL} by employing biocompatible water-immiscible ionic liquids (ILs). TMSB may be used for the synthesis of entecavir (BMS-200475).
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
3 - Flammable liquids
WGK
WGK 3
Flammpunkt (°F)
82.4 °F - closed cup
Flammpunkt (°C)
28 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Javier Velasco et al.
The Journal of organic chemistry, 78(11), 5482-5491 (2013-05-18)
Entecavir (BMS-200475) was synthesized from 4-trimethylsilyl-3-butyn-2-one and acrolein. The key features of its preparation are: (i) a stereoselective boron-aldol reaction to afford the acyclic carbon skeleton of the methylenecylopentane moiety; (ii) its cyclization by a Cp2TiCl-catalyzed intramolecular radical addition of
Wen-Yong Lou et al.
BMC biotechnology, 9, 90-90 (2009-10-24)
Whole cells are usually employed for biocatalytic reduction reactions to ensure efficient coenzyme regeneration and to avoid problems with enzyme purification and stability. The efficiency of whole cell-catalyzed bioreduction is frequently restricted by pronounced toxicity of substrate and/or product to
Bo-Bo Zhang et al.
PloS one, 7(5), e37641-e37641 (2012-06-05)
Hydrophilic ionic liquids (ILs) were employed as green solvents to construct an IL-containing co-solvent system for improving the asymmetric reduction of 4-(trimethylsilyl)-3-butyn-2-one by immobilized Candida parapsilosis cells. Among 14 hydrophilic ILs examined, 1-(2'-hydroxyl)ethyl-3-methylimidazolium nitrate (C(2)OHMIM·NO(3)) was considered as the most
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 418676-5G | 4061837581533 |
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.