Alle Fotos(2)
Wichtige Dokumente
392855
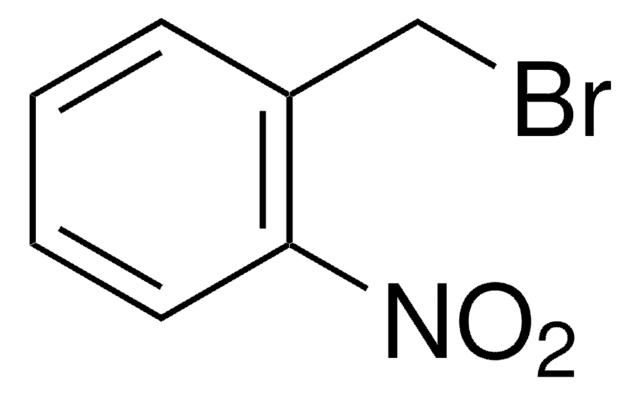
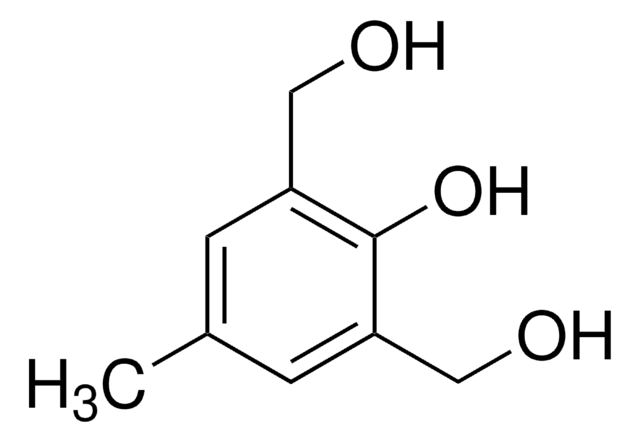
4,5-Dimethoxy-2-nitrobenzylbromid
97%
Synonym(e):
6-Nitroveratrylbromid
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Lineare Formel:
O2NC6H2(OCH3)2CH2Br
CAS-Nummer:
Molekulargewicht:
276.08
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
97%
mp (Schmelzpunkt)
131-133 °C (lit.)
Funktionelle Gruppe
bromo
nitro
SMILES String
COc1cc(CBr)c(cc1OC)[N+]([O-])=O
InChI
1S/C9H10BrNO4/c1-14-8-3-6(5-10)7(11(12)13)4-9(8)15-2/h3-4H,5H2,1-2H3
InChIKey
UEKFEYNZISYRRH-UHFFFAOYSA-N
Allgemeine Beschreibung
4,5-Dimethoxy-2-nitrobenzyl bromide (DMNBB, 1-(Bromomethyl)-4,5-dimethoxy-2-nitrobenzene) is a 4,5-dimethoxy-2-nitrobenzyl derivative. 4,5-Dimethoxy-2-nitrobenzyl (DMNB) group of DMNBB is used as a photolabile protecting group in caging technology to develop pro-drugs. Synthesis of 1-(bromomethyl)-4,5-dimethoxy-2-nitrobenzene by using 6-nitroveratraldehyde as starting reagent has been reported.
Anwendung
4,5-Dimethoxy-2-nitrobenzyl bromide (DMNB bromide, 6-nitroveratryl bromide) is suitable reagent used in the synthesis of N-(4,5-dimethoxy-2-nitrobenzyl)vanillylamine which forms caged vanilloid. It may be used in the synthesis of the following:
- 4-(4′,5′-dimethoxy-2-nitrobenzyloxy)benzaldehyde, a DMNB-caged aldehyde
- N-[4-[(4,5-dimethoxy-2-nitrobenzyl)oxy]-3-methoxybenzyl]acetamide
- caged derivative of pyridostatin ([C]-PDS)
- photosensitive polyimide (PI-DMNB)
- caged β-ecdysone
- 4-tert-butyldimethylsilyloxy-1-(2-deoxy-3,5-di-O-toluoyl-β-D-ribofuranosyl)-2-(6-nitroveratrylthio)-1H-benzimidazole, an intermediate in synthesis of phosphoramidite bearing 4-hydroxy-2-mercaptobenzimidazole (SBNV) nucleobase
- alkylation of dihydrofluorescein
- 4-(4′,5′-Dimethoxy-2′-nitrobenzyloxy)benzaldehyde
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Eye Dam. 1 - Skin Corr. 1B
Lagerklassenschlüssel
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Surface functionalization of PEEK films using photochemical routes.
Henneuse-Boxus C, et al.
Eur. Polymer J., 37(1), 9-18 (2001)
Pierre Murat et al.
Chemical communications (Cambridge, England), 49(76), 8453-8455 (2013-08-21)
The use of a caged G-quadruplex ligand allows for transcriptional control of quadruplex-containing genes using UV light as an external trigger. An important oncogene, SRC, involved in the initiation and proliferation of epithelial tumours is shown to be significantly downregulated
G Marriott et al.
Biochemistry, 35(10), 3170-3174 (1996-03-12)
An understanding of the molecular mechanism of muscle contraction will require a complete description of the kinetics of the myosin motor in vitro and in vivo. To this end chemical relaxation studies employing light-directed generation of ATP from caged ATP
Light-triggered strand exchange reaction using the change in the hydrogen bonding pattern of a nucleobase analogue.
Morihiro K, et al.
Chemical Science, 5(2), 744-750 (2014)
Synthesis and Characterizations of Positive-Working Photosensitive Polyimides Having 4, 5-Dimethoxy-o-Nitrobenzyl Side Group.
Ryu S, et al.
Bull. Korean Chem. Soc., 29(9), 1689-1689 (2008)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.