Alle Fotos(1)
Wichtige Dokumente
361267
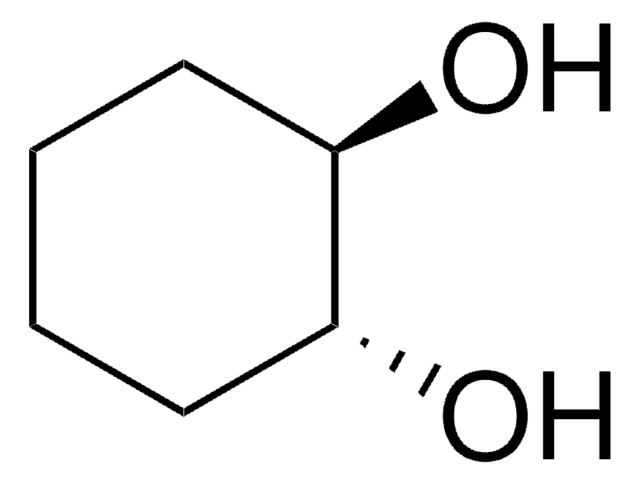
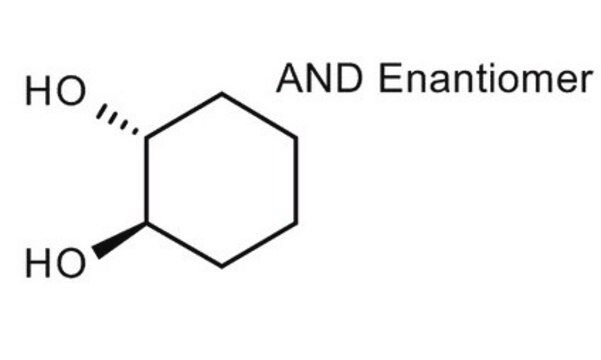
cis-1,2-Cyclohexandiol
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
C6H10(OH)2
CAS-Nummer:
Molekulargewicht:
116.16
Beilstein:
1340578
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
99%
mp (Schmelzpunkt)
97-101 °C (lit.)
Funktionelle Gruppe
hydroxyl
SMILES String
O[C@@H]1CCCC[C@@H]1O
InChI
1S/C6H12O2/c7-5-3-1-2-4-6(5)8/h5-8H,1-4H2/t5-,6+
InChIKey
PFURGBBHAOXLIO-OLQVQODUSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Core-shell-like silica nickel species nanoparticle catalyzed dehydrogenation of 1,2-cyclohexanediol to catechol is reported. Crystal structure of a Cr(V) complex with cis-1,2-cyclohexanediol is reported. Enzymatic oxidation of cis-1,2-cyclohexanediol by Gluconobacter oxydans (ATCC 621) is reported.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Bao-Hui Chen et al.
Dalton transactions (Cambridge, England : 2003), 44(3), 1023-1038 (2014-11-20)
A simple and convenient approach denoted as gel-deposition-precipitation (G-D-P) for the preparation of core-shell-like silica@nickel species nanoparticles was studied systematically. Core-shell-like silica@nickel species nanoparticles consisted of a Si-rich core and a Ni-rich shell. The G-D-P process included two steps: one
Oxidation of trans-and cis-1, 2-cyclohexanediol by Gluconobacter oxydans.
Adlercreutz P.
Applied Microbiology and Biotechnology, 30(3), 257-263 (1989)
Ruben Bartholomäus et al.
Inorganic chemistry, 52(8), 4282-4292 (2013-03-28)
The stabilization of Cr(V) by biological 1,2-diolato ligands, including carbohydrates, glycoproteins, and sialic acid derivatives, is likely to play a crucial role in the genotoxicity of Cr(VI) and has also been implicated in the antidiabetic effect of Cr(III). Previously, such
T Kiguchi et al.
Chemical & pharmaceutical bulletin, 48(10), 1536-1540 (2000-10-25)
Asymmetric spirocyclization based on intramolecular conjugate addition using a combination of a Lewis acid and an optically active cyclohexane-1,2-diol has been studied in connection with 1) the effect of substituents on the cyclohexane-1,2-diol and 2) the effect of substituents on
Ivan Liashkovich et al.
Journal of controlled release : official journal of the Controlled Release Society, 160(3), 601-608 (2012-03-06)
The efficiency of gene therapy in non-dividing cells is particularly poor due to restricted nuclear delivery rates of exogenously applied macromolecules across the nuclear pore complexes (NPCs). Therefore, improved intranuclear delivery of transgenes requires an ability to modulate the barrier
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.