Wichtige Dokumente
144894
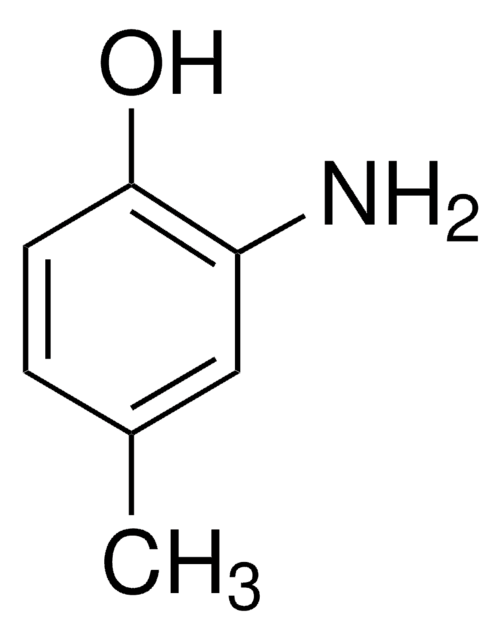
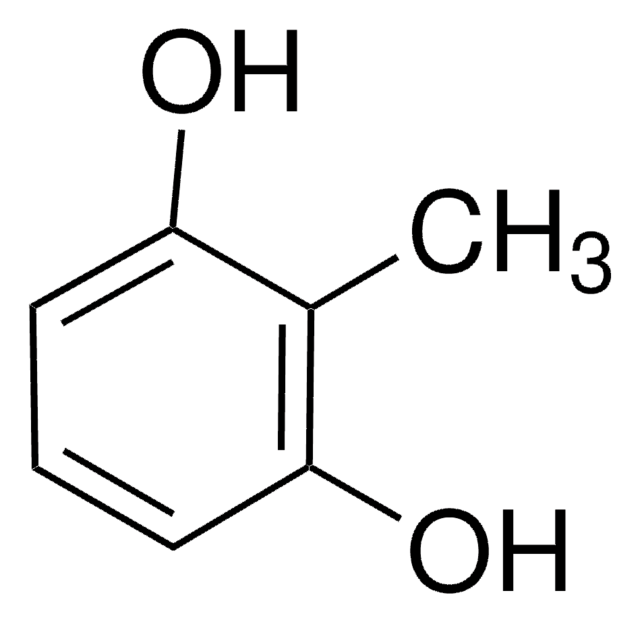
4-Amino-3-methyl-phenol
97%
Synonym(e):
2-Amino-5-hydroxy-toluol, 4-Amino-m-kresol, 4-Hydroxy-2-methyl-anilin
About This Item
Empfohlene Produkte
Assay
97%
Form
solid
mp (Schmelzpunkt)
176-179 °C (lit.)
SMILES String
Cc1cc(O)ccc1N
InChI
1S/C7H9NO/c1-5-4-6(9)2-3-7(5)8/h2-4,9H,8H2,1H3
InChIKey
QGNGOGOOPUYKMC-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Anwendung
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Skin Sens. 1A
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.