122882
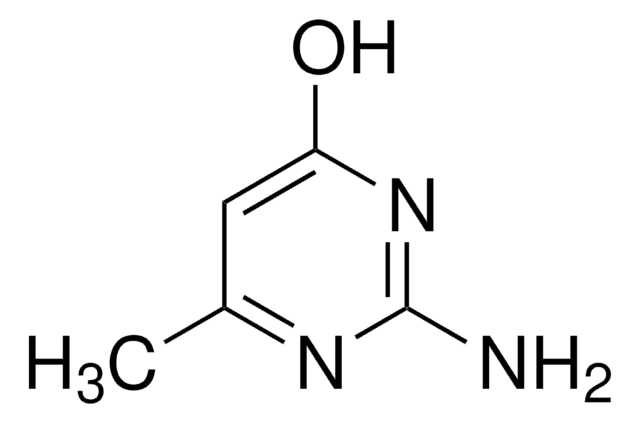
2-Amino-4-chlor-6-methylpyrimidin
97%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empirische Formel (Hill-System):
C5H6ClN3
CAS-Nummer:
Molekulargewicht:
143.57
Beilstein:
114297
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
97%
Form
solid
mp (Schmelzpunkt)
183-186 °C (lit.)
Löslichkeit
acetic acid: soluble 50 mg/mL, clear, colorless to faintly yellow
Funktionelle Gruppe
chloro
SMILES String
Cc1cc(Cl)nc(N)n1
InChI
1S/C5H6ClN3/c1-3-2-4(6)9-5(7)8-3/h2H,1H3,(H2,7,8,9)
InChIKey
NPTGVVKPLWFPPX-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
2-Amino-4-chloro-6-methylpyrimidine is a nitification inhibitor.
Anwendung
2-Amino-4-chloro-6-methylpyrimidine was used to study the influence of chlorine substitution in pyrimidine ring on proton donor ability of amino group in 2-aminopyrimidine.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Influence of chlorine-substitution in pyrimidine ring on proton donor ability in H-bond and parameters of amino group of 2-amino pyrimidine.
Borisenko VE, et al.
Vibrational Spectroscopy, 37(1), 97-109 (2005)
T Jayavarthanan et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 97, 811-824 (2012-08-21)
The solid phase FTIR and FT-Raman spectra of 2-amino-4-chloro-6-methylpyrimidine (2A4Cl6MP) have been recorded in the regions 400-4000 and 50-4,000 cm(-1), respectively. The spectra have been interpreted interms of fundamentals modes, combination and overtone bands. The structure of the molecule has
J A Hutter et al.
Biochemistry, 26(7), 1969-1973 (1987-04-07)
Thiaminase I from Bacillus thiaminolyticus strain Matsukawa et Misawa is completely and irreversibly inhibited by treatment with 4-amino-6-chloro-2-methylpyrimidine. Inhibition is a time-dependent first-order process, exhibiting a half-time of 4 h at an inhibitor concentration of 5 mM. A specific active-site-directed
Effects of nitrification inhibitors on denitrification of nitrate in soil.
Bremner JM andYeomans JC.
Biology and Fertility of Soils, 2(4), 173-179 (1986)
Christer B Aakeröy et al.
Pharmaceutics, 3(3), 601-614 (2011-01-01)
In the pharmaceutical industry, co-crystals are becoming increasingly valuable as crystalline solids that can offer altered/improved physical properties of an active pharmaceutical ingredient (API) without changing its chemical identity or biological activity. In order to identify new solid forms of
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.