1A01050
USP
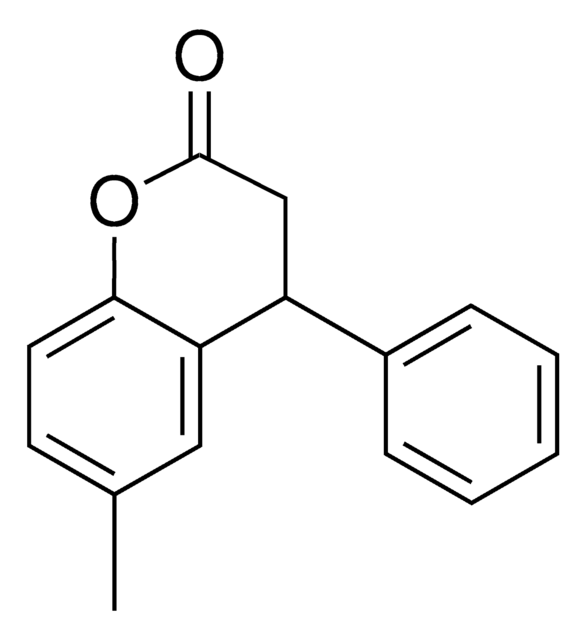
6-Methyl-4-Phenylchromanol
Pharmaceutical Analytical Impurity (PAI)
Sinonimo/i:
(6-Methyl-4-phenylchroman-2-ol)
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C16H16O2
Numero CAS:
Peso molecolare:
240.30
Codice UNSPSC:
41116100
NACRES:
NA.24
Prodotti consigliati
Grado
pharmaceutical analytical impurity (PAI)
agenzia
USP
Produttore/marchio commerciale
USP
applicazioni
pharmaceutical
Formato
neat
Temperatura di conservazione
2-8°C
Descrizione generale
[6-Methyl-4-phenylchromanol (25 mg) (6-Methyl-4-phenylchroman-2-ol)] - CAS [209747-04-6] (usp.org)6-Methyl-4-Phenylchromanol is a USP Pharmaceutical Analytical Impurity (PAI).
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Tolterodine tartrate.
Therapeutic Area: Renal.
For more information about this PAI, visit here.
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Tolterodine tartrate.
Therapeutic Area: Renal.
For more information about this PAI, visit here.
Applicazioni
6-Methyl-4-Phenylchromanol (USP PAI) is intendedfor use in analytical testing to detect, identify, and measure pharmaceuticalimpurities.
Caratteristiche e vantaggi
USP PAI advance your early analytical R&D and process development. PAI can be used in the following applications:
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.