N1895
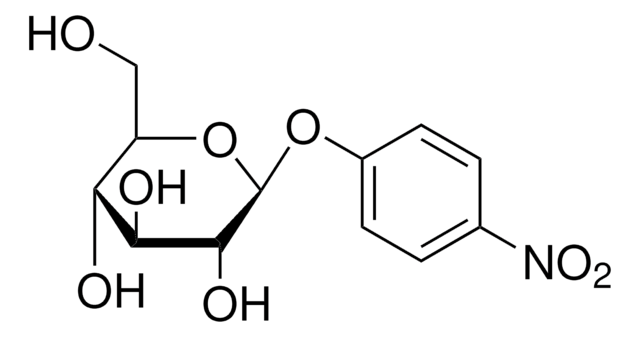
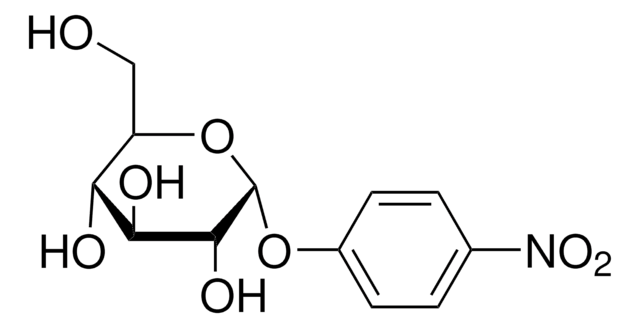
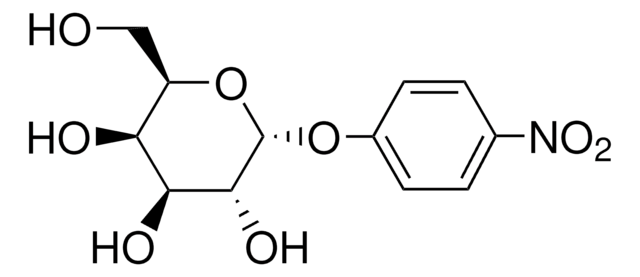
4-Nitrophenyl α-D-xylopyranoside
α-xylosidase substrate, ≥99% (HPLC), powder
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C11H13NO7
Numero CAS:
Peso molecolare:
271.22
Numero CE:
Numero MDL:
Codice UNSPSC:
12352204
ID PubChem:
NACRES:
NA.32
Prodotti consigliati
Nome del prodotto
4-Nitrophenyl α-D-xylopyranoside, α-xylosidase substrate
Saggio
≥99% (HPLC)
Stato
powder
Solubilità
methanol: soluble 20 mg/mL, clear, colorless to faintly yellow
Temperatura di conservazione
−20°C
Stringa SMILE
OC1COC(Oc2ccc(cc2)N(=O)=O)C(O)C1O
InChI
1S/C11H13NO7/c13-8-5-18-11(10(15)9(8)14)19-7-3-1-6(2-4-7)12(16)17/h1-4,8-11,13-15H,5H2
MLJYKRYCCUGBBV-UHFFFAOYSA-N
Substrati
Chromogenic substrate for α-xylosidase
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Masaki Yanagishita et al.
Kokubyo Gakkai zasshi. The Journal of the Stomatological Society, Japan, 73(1), 20-25 (2006-04-25)
Biosynthesis of proteoglycans and glycosaminoglycans in the presence of p-nitrophenyl-xyloside was studied using a primary rat ovarian granulosa cell culture system. Addition of p-nitrophenyl-xyloside into cell culture medium caused about a 700% increase of [³⁵S]sulfate incorporation (ED50 at 0.03 mM)
T Bravman et al.
FEBS letters, 495(1-2), 115-119 (2001-04-27)
A beta-xylosidase from Bacillus stearothermophilus T-6 was cloned, overexpressed in Escherichia coli and purified to homogeneity. Based on sequence alignment, the enzyme belongs to family 39 glycoside hydrolases, which itself forms part of the wider GH-A clan. The conserved Glu160
Mária Mastihubová et al.
Carbohydrate research, 339(7), 1353-1360 (2004-04-29)
Di-O-acetates and mono-O-acetates of 4-nitrophenyl beta-D-xylopyranoside were prepared by use of lipase PS-30. Polarity of organic solvents and reaction time affected the regioselectivity of the di-O-acetylation as well as the yields of monoacetates. The kinetics of acetyl groups migration in
Peter Biely et al.
Biochimica et biophysica acta, 1770(4), 565-570 (2007-01-31)
Positional specificity of NodB-like domain of a multidomain xylanase U from Clostridium thermocellum (CtAxe) was investigated. Of three monoacetates of 4-nitrophenyl beta-d-xylopyranoside the acetylxylan esterase domain showed a clear preference for the 2-acetate. Moreover, the enzyme was significantly activated by
Siyuan Li et al.
Histochemistry and cell biology, 139(1), 59-74 (2012-08-23)
Chondroitin/dermatan sulphate (CS/DS) sulphation motifs on cell and extracellular matrix proteoglycans (PGs) within stem/progenitor cell niches are involved in modulating cell phenotype during the development of many musculoskeletal connective tissues. Here, we investigate the importance of CS/DS chains and their
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.