757322
3,6-Di-tert-butylcarbazole
97%
Sinonimo/i:
3,6-Di-tert-butyl-9H-carbazole
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C20H25N
Numero CAS:
Peso molecolare:
279.42
Numero MDL:
Codice UNSPSC:
12352103
ID PubChem:
NACRES:
NA.23
Prodotti consigliati
Livello qualitativo
Saggio
97%
Stato
powder
Punto di fusione
228-233
Stringa SMILE
CC(C)(C)c1ccc2[nH]c3ccc(cc3c2c1)C(C)(C)C
InChI
1S/C20H25N/c1-19(2,3)13-7-9-17-15(11-13)16-12-14(20(4,5)6)8-10-18(16)21-17/h7-12,21H,1-6H3
OYFFSPILVQLRQA-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
3,6-Di-tert-butylcarbazole is a carbazole based material with hole transporting characteristics. The 3,6-Di-tert-butyl component of the carbazole results in an increase in the glass transition temperature (Tg) of the compound. It can be used in combination with another carbazole to form novel electroluminescent materials.
Applicazioni
3,6-Di-tert-butylcarbazole is mainly used as a monomeric precursor in the syntheses of new carbazole based materials which consist of ethynylphenyl. These materials include 9-(4-bromophenyl)-3,6-di-tert-butylcarbazol and 2-(4-(2-(4-(3,6-di-tert-butyl-9H-carbazol-9-yl)phenyl)ethynyl)benzylidene)malononitrile (PBM) which can be further be used in organic light emitting diodes (OLEDs) and optical switching devices.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Carbazole based hole transporting materials for solid state dye sensitizer solar cells: role of the methoxy groups.
Degbia M, et al.
Polymer International, 63(8), 1387-1393 (2014)
Solvent dependant optical switching in carbazole-based fluorescent nanoparticles.
Adhikari RM, et al.
Langmuir, 25(4), 2402-2406 (2009)
Synthesis and photophysical properties of carbazole-based blue light-emitting dendrimers.
Adhikari RM, et al.
The Journal of Organic Chemistry, 72(13), 4727-4732 (2007)
Bis (carbazolyl) derivatives of pyrene and tetrahydropyrene: synthesis, structures, optical properties, electrochemistry, and electroluminescence.
Kaafarani BR, et al.
Journal of Material Chemistry C, 1(8), 1638-1650 (2013)
Ji Won Yang et al.
Physical chemistry chemical physics : PCCP, 18(45), 31330-31336 (2016-11-09)
Bis(phenylsulfone) was developed as a strong electron acceptor of thermally activated delayed fluorescent emitters. The connection of two electron withdrawing phenylsulfone moieties through meta-position of phenyl produced the bis(phenylsulfone) acceptor and the strong electron acceptor strength of bis(phenylsulfone) enabled preparation
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.

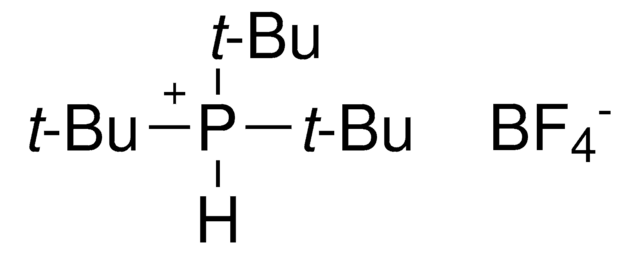
![[9-(4-Bromophenyl)]-3,6-di-tert-butyl-9H-carbazole](/deepweb/assets/sigmaaldrich/product/structures/214/779/819c00e2-ee0a-4166-977c-a7f68002b43d/640/819c00e2-ee0a-4166-977c-a7f68002b43d.png)

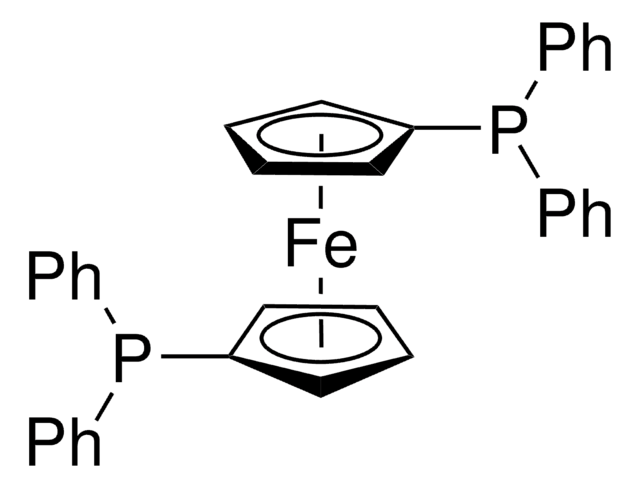
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)