559830
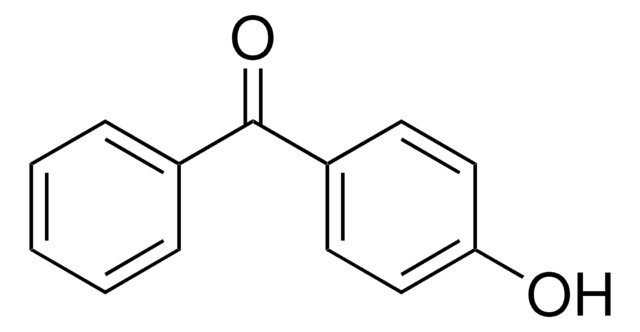
Methyl-2-benzoylbenzoate
97%
Sinonimo/i:
2-Benzoylbenzoic acid methyl ester, Methyl o-benzoylbenzoate, o-(Methoxycarbonyl)benzophenone
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C6H5COC6H4CO2CH3
Numero CAS:
Peso molecolare:
240.25
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
97%
Stato
solid
Punto di fusione
48-53 °C (lit.)
Gruppo funzionale
ester
ketone
phenyl
Stringa SMILE
COC(=O)c1ccccc1C(=O)c2ccccc2
InChI
1S/C15H12O3/c1-18-15(17)13-10-6-5-9-12(13)14(16)11-7-3-2-4-8-11/h2-10H,1H3
NQSMEZJWJJVYOI-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
Methyl-2-benzoylbenzoate is a 2-acylarylcarboxylate.
- It can undergo asymmetric transfer hydrogenation reaction in propanol in the presence of a Ruthenium catalyst.
- Methyl-2-benzoylbenzoate is formed as one of the reaction products during the reaction between methyl benzoate and lithium 2,2,6,6-tetramethylpiperidide (LiTMP) at -117°C.
- Methyl-2-benzoylbenzoate can be synthesized from the reaction between corresponding 2-substituted benzoic acid and thionyl chloride in methanol.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Aquatic Acute 1 - Aquatic Chronic 1
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Dipole stabilized carbanions. Reactions of benzoate esters with lithium 2, 2, 6, 6-tetramethylpiperidide.
Upton CJ and Beak P.
The Journal of Organic Chemistry, 40(8), 1094-1098 (1975)
One-Step Synthesis of 1-Oxo- 1,2-dihydroisoquinoline-3-carboxylic Acid Derivatives.
Nunami K, et al.
The Journal of Organic Chemistry, 44(11), 1887-1888 (1979)
Stereoselective synthesis of 3-substituted phtalides via asymmetric transfer hydrogenation using well-defined ruthenium catalysts under neutral conditions.
Everaere K, et al.
Tetrahedron Letters, 42(10), 1899-1901 (2001)
Miwa Morizane et al.
Journal of toxicology and environmental health. Part A, 78(23-24), 1450-1460 (2015-12-23)
A recent in vitro study reported that the photoinitiator 2-isopropylthioxanthone (2-ITX) is an endocrine-disrupting compound (EDC). However, it is not clear whether other photoinitiators such as 1-hydroxycyclohexyl phenyl ketone (1-HCHPK) and 2-methyl-4'-(methylthio)-2-morpholinopropiophenone (MTMP) produce endocrine-disrupting effects. The purpose of this
Miguel A Lago et al.
Food additives & contaminants. Part A, Chemistry, analysis, control, exposure & risk assessment, 33(3), 518-529 (2016-01-21)
Since the UV ink photoinitiator (PI) isopropylthioxanthone (ITX) was discovered in packaged milk, studies of print contamination have focused primarily on PIs but have also included amine synergists. Many other substances are used or formed during the print process, yet
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 559830-25G | 4061831821499 |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.