198781
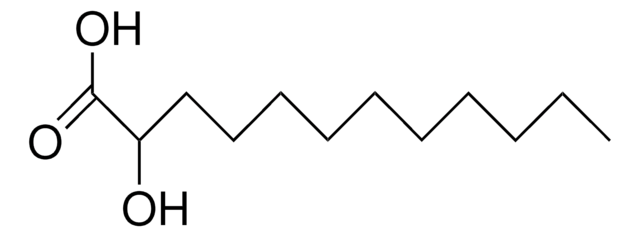
12-Hydroxydodecanoic acid
97%
Sinonimo/i:
12-Hydroxylauric acid
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
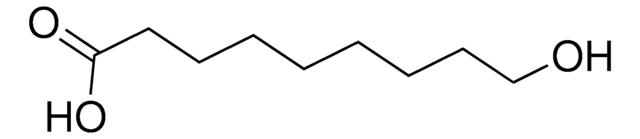
Formula condensata:
HO(CH2)11COOH
Numero CAS:
Peso molecolare:
216.32
Beilstein:
1238370
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
97%
Punto di fusione
85-88 °C (lit.)
Gruppo funzionale
carboxylic acid
hydroxyl
Stringa SMILE
OCCCCCCCCCCCC(O)=O
InChI
1S/C12H24O3/c13-11-9-7-5-3-1-2-4-6-8-10-12(14)15/h13H,1-11H2,(H,14,15)
ZDHCZVWCTKTBRY-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
12-Hydroxydodecanoic acid was used in the synthesis of high molecular weight poly[(12-hydroxydodecanoate)-co-(12-hydroxystearate)] [poly(12HD-co-12HS)] samples with variable monomer ratios using methyl 12-hydroxystearate.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
H A Dirven et al.
Journal of chromatography, 564(1), 266-271 (1991-03-08)
The formation of omega-hydroxylauric acid from lauric acid is an indicator of the activity of cytochrome P-450 IV family proteins. The two main metabolites of lauric acid, (omega-1)-and omega-hydroxylauric acid, have been completely separated by reversed-phase high-performance liquid chromatography. Measurement
P Jezek et al.
FEBS letters, 408(2), 166-170 (1997-05-19)
Fatty acid (FA) uniport via mitochondrial uncoupling protein (UcP) was detected fluorometrically with PBFI, potassium-binding benzofuran phthalate and SPQ, 6-methoxy-N-(3-sulfopropyl)-quinolinium, indicating K+ and H+, respectively. The FA structural patterns required for FA flip-flop, UcP-mediated FA uniport, activation of UcP-mediated H+
D D Giera et al.
Fundamental and applied toxicology : official journal of the Society of Toxicology, 16(2), 348-355 (1991-02-01)
Assessment of hepatic omega-oxidation of fatty acids by cytochrome P450IV enzymes in toxicology studies can be a means of evaluating test compound effects on peroxisomal proliferation. Routine assay of omega-oxidation, however, requires a simpler method of enzymatic analysis than currently
Elena Bailo et al.
Analytical and bioanalytical chemistry, 394(7), 1797-1801 (2009-06-16)
Surface-enhanced Raman scattering was used as a spectroscopic tool to investigate the changes brought upon cytochrome P450BSss after fatty acid binding. Differences in the spectra of substrate-free and substrate-bound enzyme were observed indicating the potential for this method to be
Junjun Wang et al.
International journal of molecular sciences, 19(3) (2018-03-15)
This study was aimed to clarify the effect of honokiol (Hon) on the activity of Cytochrome P450 (CYP450) enzymes, and the level of mRNA expression of liver and kidney transporters in type 2 diabetic rats induced by high-fat diet and
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.