PHR1851
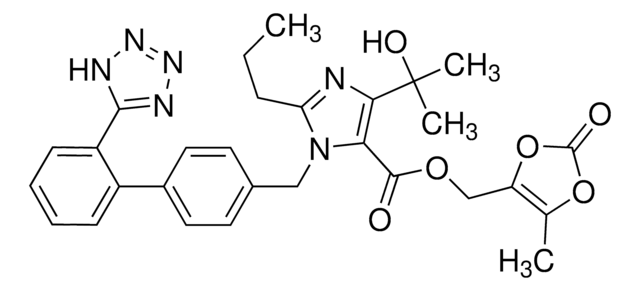
Olmesartan Medoxomil
Pharmaceutical Secondary Standard; Certified Reference Material
Synonyme(s) :
Olmesartan medoxomil
About This Item
Produits recommandés
Qualité
certified reference material
pharmaceutical secondary standard
Niveau de qualité
Agence
traceable to BP 1173
traceable to Ph. Eur. Y0001405
traceable to USP 1478367
Famille d'API
olmesartan
CofA (certificat d'analyse)
current certificate can be downloaded
Conditionnement
pkg of 200 mg
Application(s)
pharmaceutical
Format
neat
Température de stockage
-10 to -25°C
Chaîne SMILES
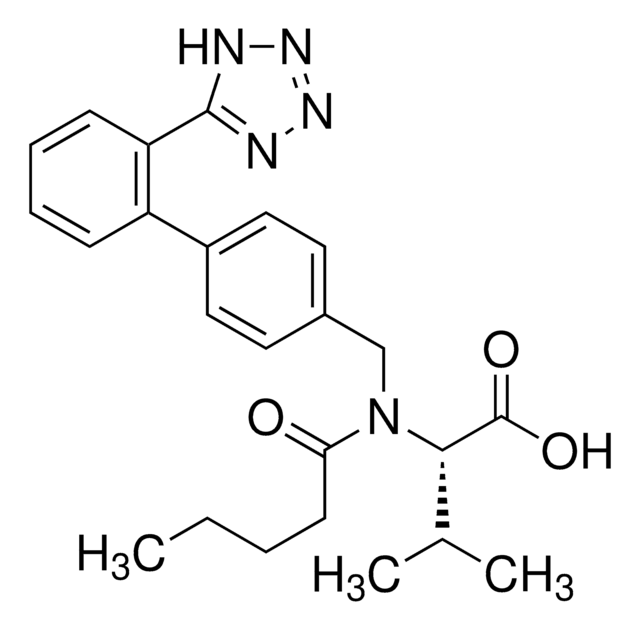
CCCC1=NC(C(O)(C)C)=C(C(OCC2=C(C)OC(O2)=O)=O)N1CC(C=C3)=CC=C3C4=CC=CC=C4C5=NN=NN5
InChI
1S/C29H30N6O6/c1-5-8-23-30-25(29(3,4)38)24(27(36)39-16-22-17(2)40-28(37)41-22)35(23)15-18-11-13-19(14-12-18)20-9-6-7-10-21(20)26-31-33-34-32-26/h6-7,9-14,38H,5,8,15-16H2,1-4H3,(H,31,32,33,34)
Clé InChI
UQGKUQLKSCSZGY-UHFFFAOYSA-N
Informations sur le gène
human ... AGTR1(185)
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Olmesartan Medoxomil is an AT1 subtype angiotensin-II receptor antagonist used in the management of hypertension. It prevents angiotensin II from binding to the AT1 receptors, thereby decreasing vasoconstriction.
Application
- Development and validation of an isocratic ultra-high performance liquid chromatography (UHPLC) based stability indicating method to determine olmesartan medoxomil and amlodipine besylate in combined tablet dosage forms
- Simultaneous determination of Olmesartan medoxomil and chlorthalidone by reversed phase-high performance liquid chromatography (RP-HPLC) in tablets
- Quantification of olmesartan medoxomil and its degradation products in bulk drugs and pharmaceutical formulations by using a systematic quality by design (QbD)-based reverse-phase liquid chromatography method
- Reversed phase-high performance liquid chromatographic (RP-HPLC) analysis of olmesartan medoxomil and hydrochlorothiazide in their combined tablet dosage form
- Determination of olmesartan medoxomil and amlodipine besylate in their combined tablet dosage form by absorption subtraction method, ratio subtraction with extended ratio subtraction method, dual wavelength technique, and second order derivative spectrophotometry
Remarque sur l'analyse
Note de bas de page
Produit(s) apparenté(s)
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
Si vous avez besoin d'assistance, veuillez contacter Service Clients
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique