31102
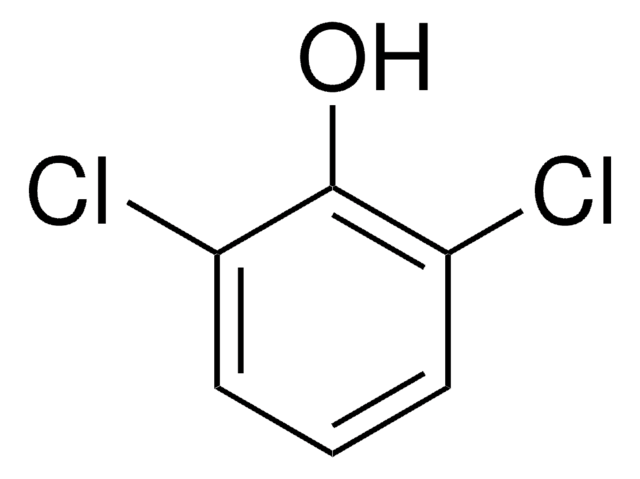
2,6-Dichlorophenol
PESTANAL®, analytical standard
About This Item
Produits recommandés
Qualité
analytical standard
Gamme de produits
PESTANAL®
Durée de conservation
limited shelf life, expiry date on the label
Technique(s)
HPLC: suitable
gas chromatography (GC): suitable
pb
218-220 °C (lit.)
Pf
64-66 °C (lit.)
Application(s)
agriculture
environmental
Format
neat
Chaîne SMILES
Oc1c(Cl)cccc1Cl
InChI
1S/C6H4Cl2O/c7-4-2-1-3-5(8)6(4)9/h1-3,9H
Clé InChI
HOLHYSJJBXSLMV-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Description générale
Application
- To study the kinetics, performance, and mechanism of the oxidative degradation of 2,6-dichlorophenol (2,6-DCP) by ferrate (VI) (Fe(VI))
- Removal of 2,6-dichlorophenol in water by copper oxide (CuO) activated peroxymonosulfate as catalyst
- Removal of 2,6-dichlorophenol by adsorption with activated polypropylene nanofiber
- Degradation of 2,6-dichlorophenol by Fe-doped titanium oxide(TiO2) sonophotocatalytic process
- Determination of 2,6-dichlorophenol by surface-enhanced Raman scattering (SERS) with molecular imprinting
Informations légales
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Skin Corr. 1B
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 2
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
Vous ne trouvez pas la bonne version ?
Si vous avez besoin d'une version particulière, vous pouvez rechercher un certificat spécifique par le numéro de lot.
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique