740683
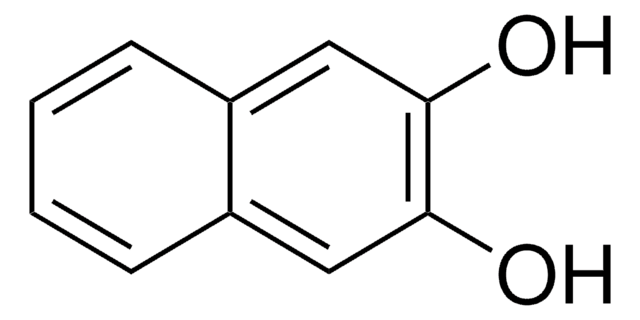
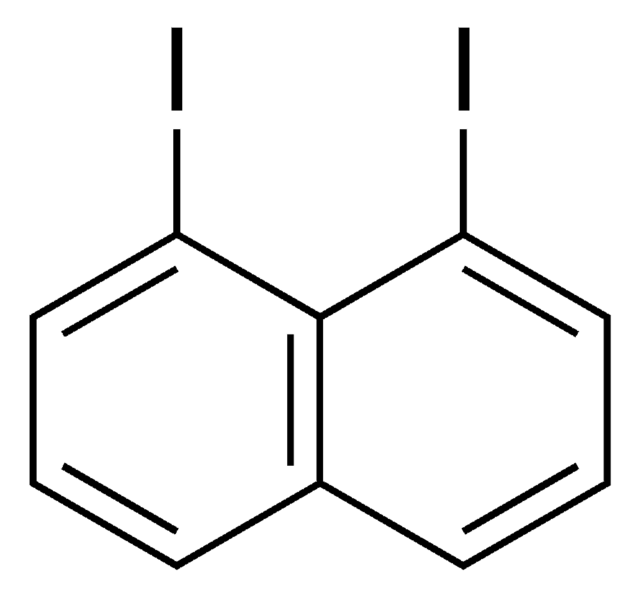
1,8-Dihydroxynaphthalene
95%
Synonyme(s) :
1,8-Naphthalenediol
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill) :
C10H8O2
Numéro CAS:
Poids moléculaire :
160.17
Beilstein:
2044947
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Niveau de qualité
Essai
95%
Forme
solid
Pf
137-143 °C
Température de stockage
2-8°C
Chaîne SMILES
Oc1cccc2cccc(O)c12
InChI
1S/C10H8O2/c11-8-5-1-3-7-4-2-6-9(12)10(7)8/h1-6,11-12H
Clé InChI
OENHRRVNRZBNNS-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Application
1,8-Dihydroxynaphthalene (DHN) can be used as:
- An intermediate in the preparation of benzo analogs of spiromamakone A.
- A starting material to synthesize naphthopyran derivatives.
- An intermediate in the total synthesis of palmarumycin CP17 analogs.
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Eye Dam. 1
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
E Thines et al.
The Journal of antibiotics, 51(4), 387-393 (1998-06-19)
From submerged cultures of Scytalidium sp. 36-93, ten metabolites were isolated due to their effects on dihydroxynaphthalene (DHN) or DOPA melanin biosynthesis. Four of the compounds, scytalols A (1a), B (1b), C (2) and D (3), are new secondary metabolites
Pigment biosynthesis and virulence.
A A Brakhage et al.
Contributions to microbiology, 2, 205-215 (1999-10-16)
Hong Jiang et al.
Gene, 602, 8-15 (2016-11-16)
A PKS1 gene responsible for the melanin biosynthesis and a NPG1 gene in Aureobasidium melanogenum XJ5-1 were cloned and characterized. An ORF of the PKS1 gene encoding a protein with 2165 amino acids contained 6495bp while an ORF of the
Shao Yu Lin et al.
Molecular plant-microbe interactions : MPMI, 25(12), 1552-1561 (2012-09-01)
Both Colletotrichum and Magnaporthe spp. develop appressoria pigmented with melanin, which is essential for fungal pathogenicity. 1,8-Dihydroxynaphthalene (1,8-DHN) is believed to be polymerized to yield melanin around the appresorial cell wall through the oxidative activity of laccases. However, no 1,8-DHN
H F Tsai et al.
The Journal of biological chemistry, 276(31), 29292-29298 (2001-05-15)
Chain lengths and cyclization patterns of microbial polyketides are generally determined by polyketide synthases alone. Fungal polyketide melanins are often derived from a pentaketide 1,8-dihydroxynaphthalene, and pentaketide synthases are used for synthesis of the upstream pentaketide precursor, 1,3,6,8-tetrahydroxynaphthalene (1,3,6,8-THN). However
Global Trade Item Number
| Référence | GTIN |
|---|---|
| 740683-1G | 4061832884394 |
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique