Wichtige Dokumente
913294
keYPhos™
Umicore
Synonym(e):
CyYPhos(Me)PCy2, Tricyclohexyl(1-(dicyclohexyl-phosphanyl)ethylidene)-phosphane
About This Item
Empfohlene Produkte
Produktbezeichnung
keYPhos™,
Form
powder
Qualitätsniveau
Eignung der Reaktion
reagent type: ligand
mp (Schmelzpunkt)
167-169 °C
Funktionelle Gruppe
phosphine
SMILES String
[P](=C(P(C5CCCCC5)C4CCCCC4)C)(C3CCCCC3)(C2CCCCC2)C1CCCCC1
InChI
1S/C32H58P2/c1-27(33(28-17-7-2-8-18-28)29-19-9-3-10-20-29)34(30-21-11-4-12-22-30,31-23-13-5-14-24-31)32-25-15-6-16-26-32/h28-32H,2-26H2,1H3
InChIKey
TZVGFYHCFCNLKS-UHFFFAOYSA-N
Allgemeine Beschreibung
Anwendung
Learn more about ylide-functionalized phosphines (YPhos)
Leistungsmerkmale und Vorteile
Rechtliche Hinweise
This product, its manufacturing or use, is the subject of one or more issued or pending U.S. Patents (and foreign equivalents) owned or controlled by Umicore PMC. The purchase of this product from Umicore PMC through Sigma-Aldrich, its affiliates or their authorized distributors conveys to the buyer a limited, one-time, non-exclusive, non-transferable, non-assignable license. Buyer′s use of this product may infringe patents owned or controlled by third parties. It is the sole responsibility of buyer to ensure that its use of the product does not infringe the patent rights of third parties or exceed the scope of the license granted herein.
For any further information on product please refer to your local Umicore PMC contact at www.pmc.umicore.com.
Ähnliches Produkt
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumente section.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.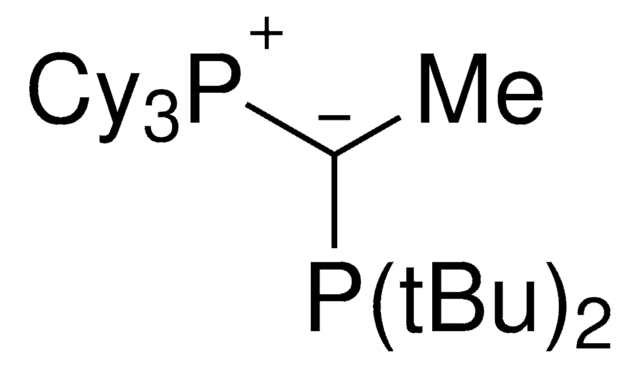
![[1,1′-Bis(diphenylphosphino)ferrocen]dichlorpalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)


![Dichloro[2,2′-bis(diphenylphosphino)-1,1′-binaphthyl]palladium(II) 97%](/deepweb/assets/sigmaaldrich/product/structures/351/904/a38f7a0d-b8a0-448f-b949-4b983e7f1eb3/640/a38f7a0d-b8a0-448f-b949-4b983e7f1eb3.png)