513830
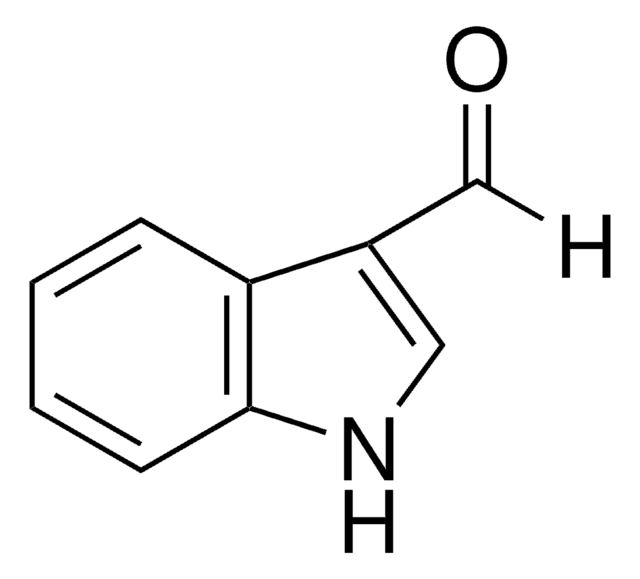
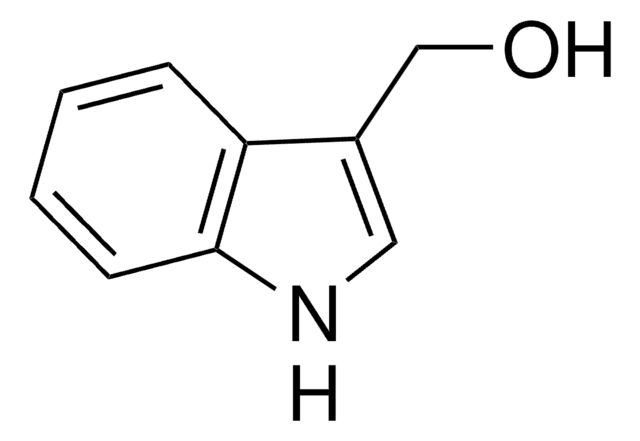
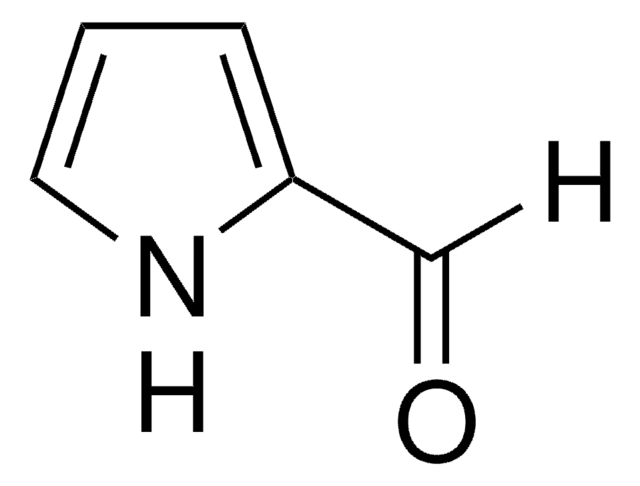
Indol-5-carboxaldehyd
98%
Synonym(e):
5-Formylindole
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empirische Formel (Hill-System):
C9H7NO
CAS-Nummer:
Molekulargewicht:
145.16
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
98%
mp (Schmelzpunkt)
100-103 °C (lit.)
Funktionelle Gruppe
aldehyde
SMILES String
O=Cc1ccc2[nH]ccc2c1
InChI
1S/C9H7NO/c11-6-7-1-2-9-8(5-7)3-4-10-9/h1-6,10H
InChIKey
ADZUEEUKBYCSEY-UHFFFAOYSA-N
Verwandte Kategorien
Anwendung
Indole-5-carboxaldehyde can be used as a reactant in the:
- Preparation of curcumin derivatives as anti-proliferative & anti-inflammatory agents
- Preparation of analogs of botulinum neurotoxin serotype A protease inhibitors
- Stereoselective synthesis of dibenzylideneacetone derivatives as β-amyloid imaging probes
- Synthesis of para-para stilbenophanes by McMurry coupling
- Stereoselective synthesis of heteroaromatic (E)-α,β-unsaturated ketones from aldehydes
- Structure-based drug design of aurora kinase A inhibitors
- Preparation of 5-indolyl linked 15- and 18-membered azacrown ethers to study their cation-π interactions.
- Preparation of bibenzimidazole derivatives substituted 5-indolyl moiety in the study of inhibition of topoisomerase I activity.
- Synthesis of (5-(4-(3,4,5-trimethoxybenzoyl)-1H-imidazol-2-yl)-1H-indol-2-yl)(3,4,5-trimethoxyphenyl)methanone and radioiodinated indolochalcone.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Raquel Álvarez et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 17(12), 3406-3419 (2011-02-24)
The synthesis of a new family of methoxy-substituted [2.7]- and [2.8]paracyclophanes linked by 3-oxapentamethylene-1,5-dioxy and hexamethylene-1,6-dioxy bridges has been carried out by using the McMurry methodology. Related indole compounds were also synthesised. Olefin-to-diol ratios depended on the bridge length, the
Balducci, E.; et al.
European Journal of Organic Chemistry, 311-311 (2011)
Houlihan WJ.
The Chemistry of Heterocyclic Compounds, 367-367 (2009)
Mohane Selvaraj Coumar et al.
Journal of medicinal chemistry, 52(4), 1050-1062 (2009-01-15)
Aurora kinases have emerged as attractive targets for the design of anticancer drugs. Through structure-based virtual screening, novel pyrazole hit 8a was identified as Aurora kinase A inhibitor (IC(50) = 15.1 microM). X-ray cocrystal structure of 8a in complex with
Petr Capek et al.
ACS chemical neuroscience, 2(6), 288-293 (2011-07-12)
Botulinum neurotoxin (BoNT), the etiological agent that causes the neuroparalytic disease botulism, has become a highly studied drug target in light of the potential abuse of this toxin as a weapon of bioterrorism. In particular, small molecule inhibitors of the
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.