Alle Fotos(2)
Wichtige Dokumente
419648
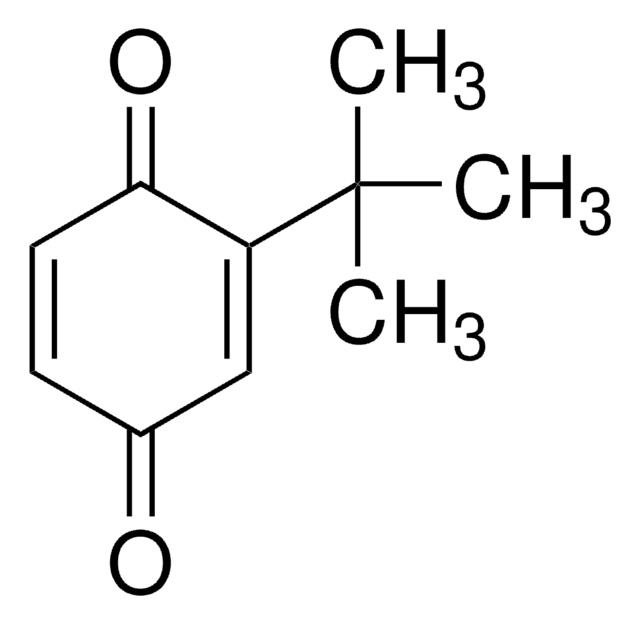
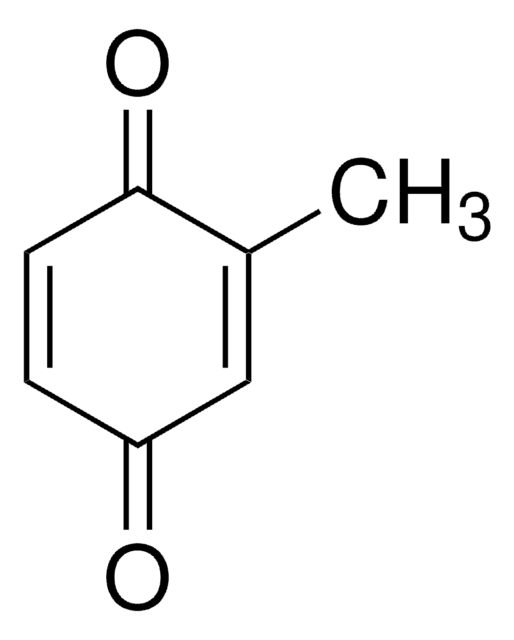
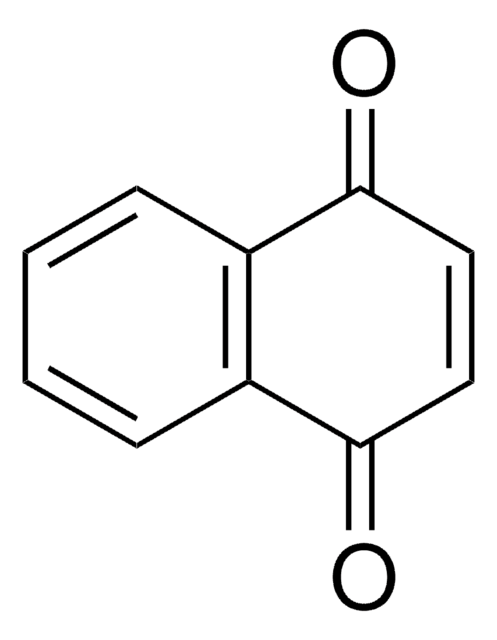
2,5-Di-tert-butyl-1,4-benzochinon
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Lineare Formel:
[(CH3)3C]2C6H2(=O)2
CAS-Nummer:
Molekulargewicht:
220.31
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
99%
mp (Schmelzpunkt)
152-154 °C (lit.)
Funktionelle Gruppe
ketone
SMILES String
CC(C)(C)C1=CC(=O)C(=CC1=O)C(C)(C)C
InChI
1S/C14H20O2/c1-13(2,3)9-7-12(16)10(8-11(9)15)14(4,5)6/h7-8H,1-6H3
InChIKey
ZZYASVWWDLJXIM-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
2,5-Di-tert-butyl-1,4-benzoquinone (DTBBQ) is an 2,5-disubstituted quinone. It is an antibacterial compound. It has been isolated from marine Streptomyces sp. VITVSK1. Pressure dependance on the intramolecular and intermolecular migration rates of Na+ and K+ in a 2,5-di-tert-butyl-1,4-benzoquinone ion pair have been evaluated by using a high-pressure EPR technique.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Catalytic aerobic deamination of activated primary amines by a model for the quinone cofactor of mammalian copper amine oxidases.
L M Sayre et al.
Methods in enzymology, 258, 53-69 (1995-01-01)
L C Rome et al.
The Journal of physiology, 526 Pt 2, 279-286 (2000-07-15)
1. The rate at which an isometrically contracting muscle uses energy is thought to be proportional to its twitch speed. In both slow and fast muscles, however, a constant proportion (25-40 %) of the total energy has been found to
Vinay Gopal Jannu et al.
International journal of bioinformatics research and applications, 11(2), 142-152 (2015-03-20)
The incidence of bacterial disease has increased tremendously in the last decade, because of the emergence of drug resistance strains within the bacterial pathogens. The present study was to investigate the antibacterial compound 2,5-di-tert-butyl-1,4-benzoquinone (DTBBQ) isolated from marine Streptomyces sp.
L Missiaen et al.
European journal of pharmacology, 227(4), 391-394 (1992-12-01)
Specific inhibitors of the endoplasmic-reticulum Ca2+ pump will deplete intracellular stores and are therefore useful to study the role of store depletion on plasma-membrane Ca2+ permeability. We now report that the Ca(2+)-pump inhibitor 2,5-di-(tert-butyl)-1,4-benzohydroquinone (tBuBHQ) reduces the passive Ca2+ leak
R J Dolor et al.
The American journal of physiology, 262(1 Pt 1), C171-C181 (1992-01-01)
We have investigated the role of the intracellular Ca2+ pool in regulating Ca2+ entry into vascular endothelial cells. The intracellular Ca2+ pool was mobilized using either thapsigargin (TG) or 2',5'-di(tert-butyl)-1,4-benzohydroquinone (BHQ), inhibitors of the endoplasmic reticulum Ca(2+)-adenosinetriphosphatase (ATPase). Mobilization of
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.