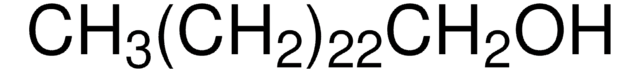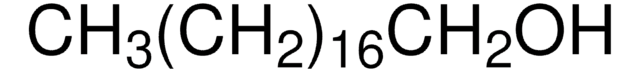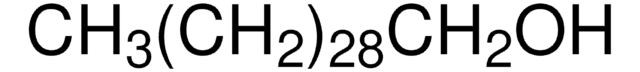Alle Fotos(1)
Wichtige Dokumente
169102
1-Docosanol
98%
Synonym(e):
Behenylalkohol
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
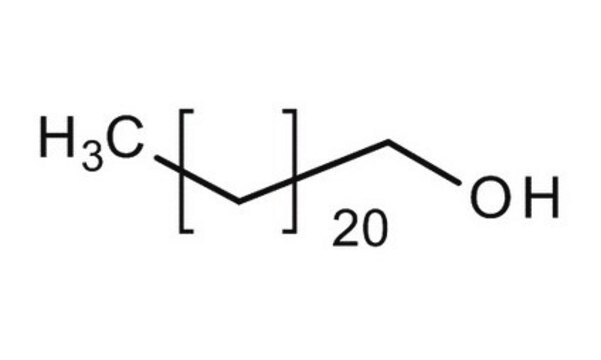
Lineare Formel:
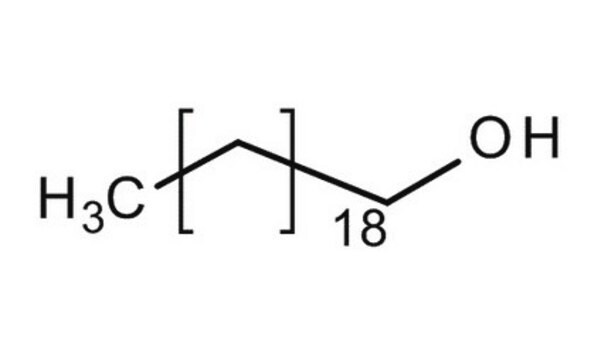
CH3(CH2)21OH
CAS-Nummer:
Molekulargewicht:
326.60
Beilstein:
1770470
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
98%
bp
180 °C/0.22 mmHg (lit.)
mp (Schmelzpunkt)
65-72 °C (lit.)
Funktionelle Gruppe
hydroxyl
SMILES String
CCCCCCCCCCCCCCCCCCCCCCO
InChI
1S/C22H46O/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22-23/h23H,2-22H2,1H3
InChIKey
NOPFSRXAKWQILS-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
1-Docosanol inhibits replication of certain viruses (herpes simplex virus and respiratory syncytial virus) within primary target cells in vitro. It has been isolated from Clematis brevicaudata.
Anwendung
1-Docosanol was used in the synthesis of series of amphiphilic dendrimers with hydrophilic aliphatic polyether-type dendritic core and hydrophobic docosyl peripheries.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
nwg
Flammpunkt (°F)
410.0 °F
Flammpunkt (°C)
210 °C
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Ai-Mei Yang et al.
Zhong yao cai = Zhongyaocai = Journal of Chinese medicinal materials, 32(10), 1534-1537 (2010-02-02)
To study the chemical constituents from Clematis brevicaudata. The compounds were isolated by column chromatography and their structures were elucidated through spectroscopic analysis (NMR). Eight compounds were isolated and identified as: palmitic acid (1), 1-docosanol (2), pentacosanoic acid-2', 3'-dihydroxypropyl ester
Synthesis and self-assembly of amphiphilic dendrimers based on aliphatic polyether-type dendritic cores.
Cho B-K, et al.
Macromolecules, 37(11), 4227-4234 (2004)
Antiviral activity of 1-docosanol, an inhibitor of lipid-enveloped viruses including herpes simplex.
D H Katz et al.
Proceedings of the National Academy of Sciences of the United States of America, 88(23), 10825-10829 (1991-12-01)
This article reports that 1-docosanol, a 22-carbon-long saturated alcohol, exerts a substantial inhibitory effect on replication of certain viruses (e.g., herpes simplex virus and respiratory syncytial virus) within primary target cells in vitro. To study the basis for its viral
John F Marcelletti
Antiviral research, 56(2), 153-166 (2002-10-09)
Interactions between docosanol (n-docosanol, behenyl alcohol) and nucleoside or pyrophosphate analogs were investigated in vitro. The anti-HSV activity of acyclovir (ACV) was synergistically enhanced by treatment of cells with docosanol as judged by inhibition of progeny virus production and plaque
Clara L Shaw et al.
Journal of chemical ecology, 37(4), 329-339 (2011-03-23)
The uropygial secretions of some bird species contain volatile and semivolatile compounds that are hypothesized to serve as chemical signals. The abundance of secretion components varies with age and season, although these effects have not been investigated in many species.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.