302589
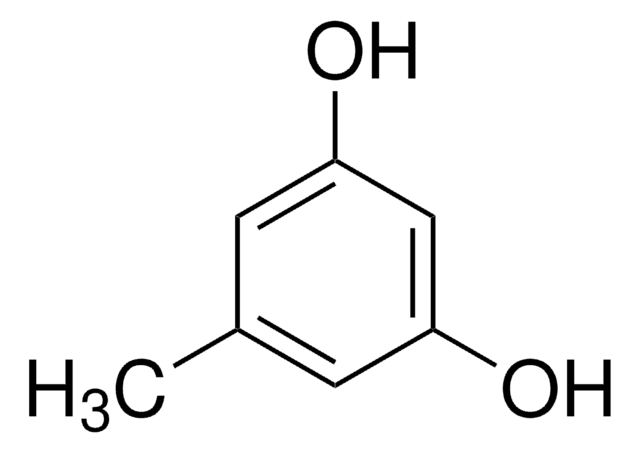
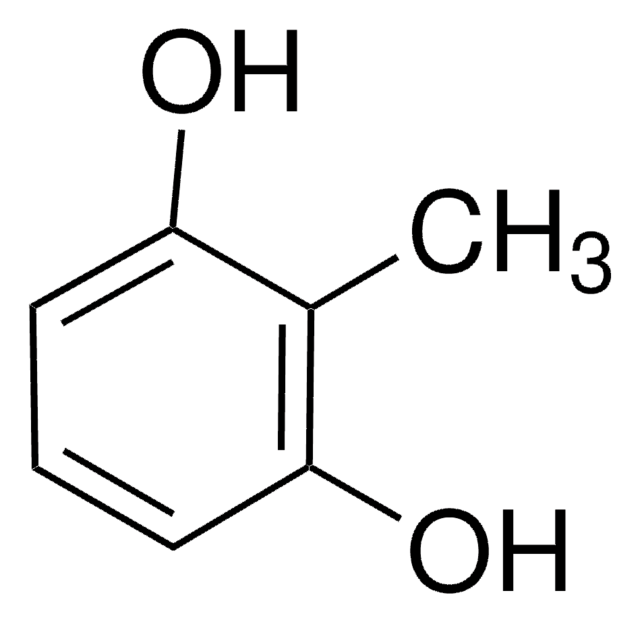
2-Methylresorcinol
98%
Synonyme(s) :
2,6-Dihydroxytoluene
About This Item
Produits recommandés
Niveau de qualité
Essai
98%
Forme
solid
pb
264 °C (lit.)
Pf
114-120 °C (lit.)
Chaîne SMILES
Cc1c(O)cccc1O
InChI
1S/C7H8O2/c1-5-6(8)3-2-4-7(5)9/h2-4,8-9H,1H3
Clé InChI
ZTMADXFOCUXMJE-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Description générale
Application
- C-5-bromo-2-hydroxyphenylcalix[4]-2-methylresorcinarene
- tripyrrane analogs
- series of novel aromatic benziporphyrins
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 3 Oral - Aquatic Acute 1 - Eye Dam. 1 - Skin Sens. 1B
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
275.0 °F - closed cup
Point d'éclair (°C)
135 °C - closed cup
Équipement de protection individuelle
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Protocoles
Separation of Resorcinol 50 mg/mL; Pyrocatechol; 2-Methylresorcinol; 4-Methylcatechol; 2,5-Dimethylresorcinol 50 mg/mL; 3-Methylcatechol 50 mg/mL; 4-Nitrocatechol 50 mg/mL
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique