C0267
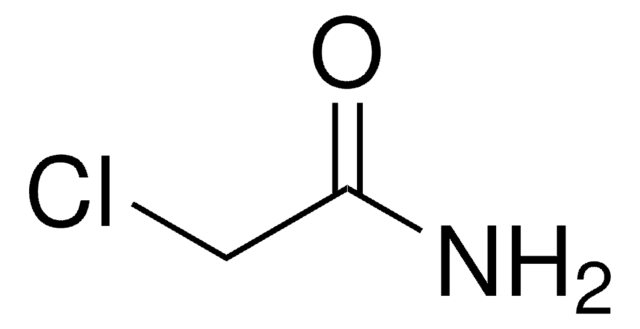
2-Chloroacetamide
≥98%
Synonym(s):
Chloracetamide, alpha-Chloroacetamide
About This Item
Recommended Products
vapor pressure
0.05 mmHg ( 20 °C)
Assay
≥98%
form
powder
mp
116-118 °C (lit.)
SMILES string
NC(=O)CCl
InChI
1S/C2H4ClNO/c3-1-2(4)5/h1H2,(H2,4,5)
InChI key
VXIVSQZSERGHQP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- 2-Amino-4-chloroquinolines via o-alkylation of 4-chloro-1H-quinolin-2-ones and subsequent Smiles rearrangement.
- 2-(Formylaryloxy)acetamides by reacting with hydroxybenzaldehydes in the presence of potassium carbonate as a base.
- Biarylamine derivatives via aluminum triflate-catalyzed direct amination of benzhydrols.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Repr. 2 - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 2
Flash Point(F)
338.0 °F
Flash Point(C)
170 °C
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
In this study, we developed a rapid trypsin digest kit that, at elevated temperatures, yielded reliable, reproducible results in less than 2 hours on a wide variety of substrates for mass spectrometry.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service