159417
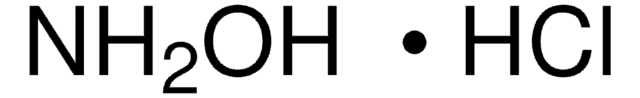
Hydroxylamine hydrochloride
ReagentPlus®, 99%
Synonyme(s) :
Hydroxylammonium chloride
About This Item
Produits recommandés
Pression de vapeur
0.001 hPa ( 50 °C)
Niveau de qualité
Gamme de produits
ReagentPlus®
Essai
99%
Forme
crystalline
Technique(s)
inhibition assay: suitable
pH
2.5-3.5 (20 °C, 50 g/L)
Pf
155-157 °C (dec.) (lit.)
Densité
1.67 g/mL at 25 °C (lit.)
Chaîne SMILES
Cl.NO
InChI
1S/ClH.H3NO/c;1-2/h1H;2H,1H2
Clé InChI
WTDHULULXKLSOZ-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- in the synthesis of primary amides from aldehydes in the presence of cesium carbonate (Cs2CO3) as a catalyst.
- in the conversion of alicyclic /aliphatic carbonyl compounds and the aromatic aldehydes into corresponding oximes.
- in the one-pot synthesis of nitriles from aldehydes in the presence of sodium sulfate (anhyd) and sodium bicarbonate catalysts.
- It can also be used as a reducing agent in the preparation of single-layer reduced graphene oxide (RGO) sheets and films.
Actions biochimiques/physiologiques
Informations légales
Vous ne trouvez pas le bon produit ?
Essayez notre Outil de sélection de produits.
Mention d'avertissement
Warning
Mentions de danger
Classification des risques
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 2 - Carc. 2 - Eye Irrit. 2 - Met. Corr. 1 - Skin Irrit. 2 - Skin Sens. 1 - STOT RE 2 Oral
Organes cibles
spleen
Code de la classe de stockage
4.1A - Other explosive hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique