37018
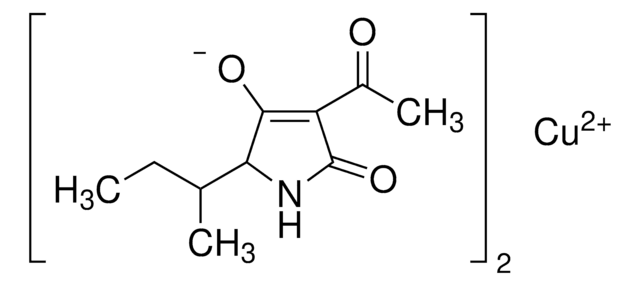
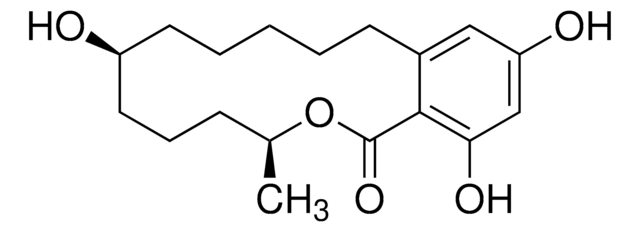
Tenuazonic acid
analytical standard
Synonyme(s) :
(5S)-3-Acetyl-1,5-dihydro-4-hydroxy-5-[(1S)-1-methylpropyl]-2H-pyrrol-2-one, (S)-3-Acetyl-5-(S)-sec-butyltetramic acid, (S)-3-Acetyl-5-sec-butyl-4-hydroxy-3-pyrrolin-2-one
About This Item
Produits recommandés
Qualité
analytical standard
Niveau de qualité
Durée de conservation
limited shelf life, expiry date on the label
Technique(s)
HPLC: suitable
gas chromatography (GC): suitable
Application(s)
cleaning products
cosmetics
food and beverages
personal care
Format
neat
Température de stockage
−20°C
Chaîne SMILES
CC[C@H](C)[C@@H]1NC(=O)C(C(C)=O)=C1O
InChI
1S/C10H15NO3/c1-4-5(2)8-9(13)7(6(3)12)10(14)11-8/h5,8,13H,4H2,1-3H3,(H,11,14)/t5-,8-/m0/s1
Clé InChI
CEIZFXOZIQNICU-XNCJUZBTSA-N
Description générale
Application
- Cornflakes using high-performance liquid chromatography (HPLC) technique.
- Human urine samples using isotope dilution assay method and liquid chromatography coupled to a hybrid triple quadrupole/linear ion trap mass spectrometer.
- Cereals using high-performance liquid chromatography–electrospray ionization ion-trap multistage mass spectrometry (HPLC- ESI ion-trap (IT) MS2). ESI Fourier transform-ion cyclotron resonance tandem mass spectrometry (FTICR-MS2) technique is used for product characterization, post derivatization with 2,4-dinitrophenylhydrazine.
Reconstitution
Remarque sur l'analyse
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 3 Oral
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique