522856
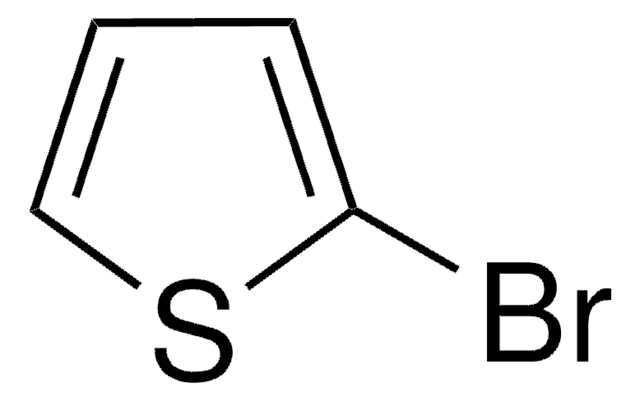
5-Bromo-2,2′-bithiophene
96%
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill):
C8H5BrS2
Numéro CAS:
Poids moléculaire :
245.16
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Pureté
96%
Forme
solid
Pf
29-32 °C (lit.)
Chaîne SMILES
Brc1ccc(s1)-c2cccs2
InChI
1S/C8H5BrS2/c9-8-4-3-7(11-8)6-2-1-5-10-6/h1-5H
Clé InChI
OMOAIGVIYUXYAU-UHFFFAOYSA-N
Description générale
5-Bromo-2,2′-bithiophene is a bromothiophene derivative. Its reaction with various aryl iodides bearing an electron-donating or electron-withdrawing substituent has been described. It can be synthesized from 2,2′-bithiophene.
Application
5-Bromo-2,2′-bithiophene may be used in the synthesis of trimethyl-[2,2′;5′,2″;5″,2″]quaterthiophen-5-yl-silane (4TTMS) and 5-hexylsulfanyl-2,2′:5′,2′′-terthiophene.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Mark E Roberts et al.
Proceedings of the National Academy of Sciences of the United States of America, 105(34), 12134-12139 (2008-08-20)
The development of low-cost, reliable sensors will rely on devices capable of converting an analyte binding event to an easily read electrical signal. Organic thin-film transistors (OTFTs) are ideal for inexpensive, single-use chemical or biological sensors because of their compatibility
Kei Kobayashi et al.
Organic letters, 7(22), 5083-5085 (2005-10-21)
[reaction: see text] Bromothiophene derivatives react with aryl iodides catalyzed by a palladium complex in the presence of a silver(I) nitrate/potassium fluoride system to induce coupling at the C-H bond, while the carbon-bromine bond is intact. The produced coupling product
Silole-Containing. pi.-Conjugated Systems. 3.1 A Series of Silole-Thiophene Cooligomers and Copolymers: Synthesis, Properties, and Electronic Structures.
Tamao K, et al.
Macromolecules, 28(25), 8668-8675 (1995)
Three-dimensional tetra (oligothienyl) silanes as donor material for organic solar cells.
Roquet S, et al.
Journal of Materials Chemistry, 16(29), 3040-3045 (2006)
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique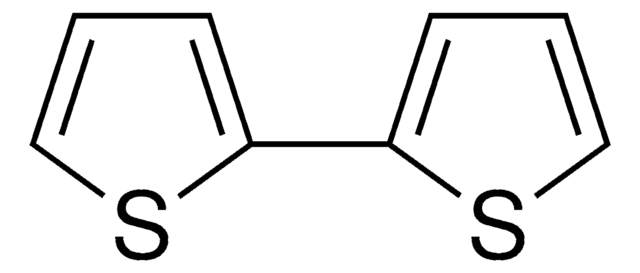

![Benzo[1,2-b:4,5-b′]dithiophene-4,8-dione 97%](/deepweb/assets/sigmaaldrich/product/structures/418/544/b7faac0b-ad09-4b42-a9fa-aeb38017a39e/640/b7faac0b-ad09-4b42-a9fa-aeb38017a39e.png)
![Thieno[3,2-b]thiophene 95%](/deepweb/assets/sigmaaldrich/product/structures/353/609/429fd4bf-e217-4371-80a3-9e5a4d88908b/640/429fd4bf-e217-4371-80a3-9e5a4d88908b.png)
![2,5-Bis(trimethylstannyl)-thieno[3,2-b]thiophene 97%](/deepweb/assets/sigmaaldrich/product/structures/126/532/26557e94-858e-4c96-90de-ca88d84a8727/640/26557e94-858e-4c96-90de-ca88d84a8727.png)