429074
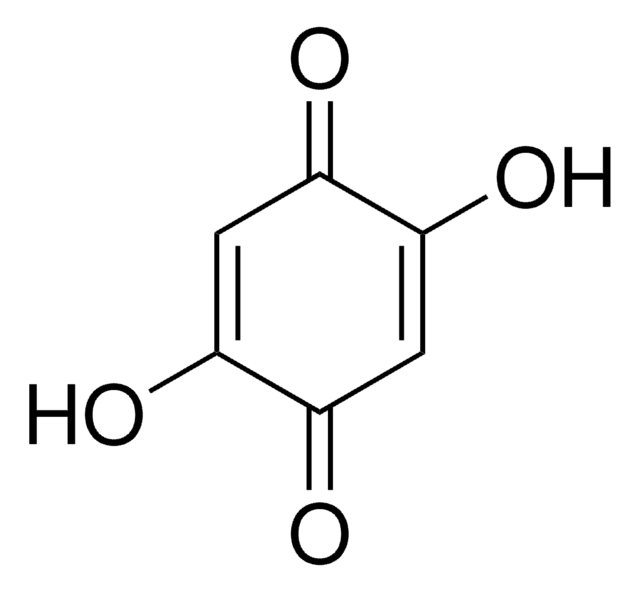
2-tert-Butyl-1,4-benzoquinone
98%
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule linéaire :
(CH3)3CC6H3(=O)2
Numéro CAS:
Poids moléculaire :
164.20
Numéro CE :
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Pureté
98%
Forme
solid
Pf
54-58 °C (lit.)
Groupe fonctionnel
ketone
Chaîne SMILES
CC(C)(C)C1=CC(=O)C=CC1=O
InChI
1S/C10H12O2/c1-10(2,3)8-6-7(11)4-5-9(8)12/h4-6H,1-3H3
Clé InChI
NCCTVAJNFXYWTM-UHFFFAOYSA-N
Description générale
2-tert-Butyl-1,4-benzoquinone (TBQ, TBBQ, tBQ, BuBQ, BQ , tert-butyl-p-quinone) is a 1,4-benzoquinone derivative. It is a major metabolite of the food additive, butylated hydroxyanisole (BHA). TBQ is reported to be strongly cytotoxic in human monocytic leukemia U937 cells. TBQ is an oxidation product of 2-tert-butylhydroquinone (TBHQ). Studies confirm that TBQ induces apoptosis and cell proliferation inhibition in chronic myelogenous leukemia (CML) cells. Its binding interactions with lysozyme has been examined and found to be intermediate between BHA and TBHQ. It has been reported to be synthesized by the titanium superoxide catalyzed oxidation of 2-tert-butylphenol using aq. 30% H2O2. TBQ is one of the main neoformed compounds from TBHQ decomposition in PLA-TBHQ film (Poly lactic acid).
Application
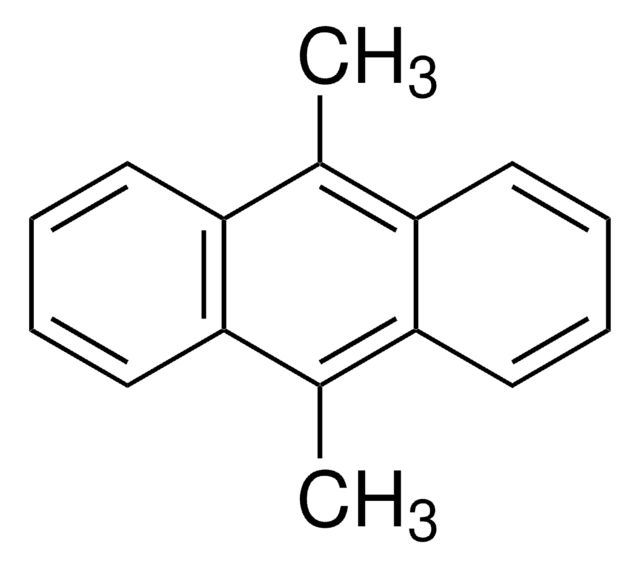
2-tert-Butyl-1,4-benzoquinone may be used in the synthesis of azatrioxa[8]circulene.
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
2-tert-butyl-1, 4-benzoquinone Induces Apoptosis in Chronic Myeloid Leukemia Cells Resistant to Imatinib via Inducing Caspase-Dependent Bcr-Abl Downregulation.
Shi X, et al.
Medicinal Chemistry, 4, 784-790 (2014)
R Kahl et al.
Toxicology, 59(2), 179-194 (1989-12-01)
The synthetic antioxidant butylated hydroxyanisole (BHA) stimulates superoxide formation in rat liver microsomes up to 10-fold. This stimulation is prevented by the monooxygenase inhibitor metyrapone and does not occur when NADH is consumed instead of NADPH indicating that metabolic activation
W H Kalus et al.
Environmental health perspectives, 102(1), 96-99 (1994-01-01)
We examined t-butylhydroquinone (t-BHQ) and t-butylquinone (t-BuQ), two of the major microsomal metabolites of the synthetic antioxidant butylated hydroxyanisole (BHA), for their ability to react with the xenobiotic arylamines aniline and N-methylaniline. A number of substances were isolated by thin-layer
Azatrioxa [8] circulenes: Planar Anti-Aromatic Cyclooctatetraenes.
Nielsen CB, et al.
Chemistry (Weinheim An Der Bergstrasse, Germany), 19(12), 3898-3904 (2013)
P A Schilderman et al.
Carcinogenesis, 16(3), 507-512 (1995-03-01)
The food additive butylated hydroxyanisole (BHA) has been shown to induce gastrointestinal hyperplasia in rodents by an unknown mechanism. The relevance of this observation for human risk assessment is not clear. We therefore analysed the effect of BHA and its
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique