T5648
Tamoxifen
≥99% (TLC), powder, PKC inhibitor
Synonym(s):
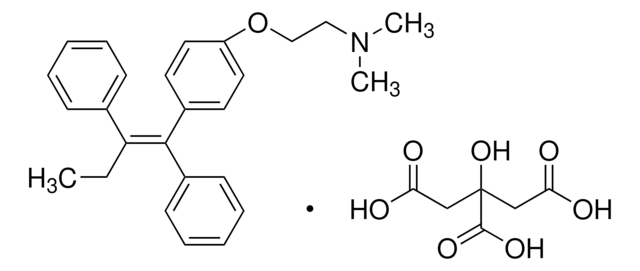
trans-Tamoxifen, (Z)-1-(p-Dimethylaminoethoxyphenyl)-1,2-diphenyl-1-butene, trans-2-[4-(1,2-Diphenyl-1-butenyl)phenoxy]-N,N-dimethylethylamine
About This Item
Recommended Products
product name
Tamoxifen, ≥99%
sterility
(Not packaged in a sterile environment)
Assay
≥99%
mp
97-98 °C (lit.)
solubility
H2O: insoluble <0.1% at 20 °C
chloroform: soluble 50 mg/mL, clear, colorless to faintly yellow
2-propanol: soluble
DMSO: soluble
ethanol: soluble
methanol: soluble
propylene glycol: soluble
antibiotic activity spectrum
neoplastics
Mode of action
enzyme | inhibits
originator
AstraZeneca
storage temp.
2-8°C
SMILES string
CC\C(c1ccccc1)=C(/c2ccccc2)c3ccc(OCCN(C)C)cc3
InChI
1S/C26H29NO/c1-4-25(21-11-7-5-8-12-21)26(22-13-9-6-10-14-22)23-15-17-24(18-16-23)28-20-19-27(2)3/h5-18H,4,19-20H2,1-3H3/b26-25-
InChI key
NKANXQFJJICGDU-QPLCGJKRSA-N
Gene Information
human ... CYP1A2(1544) , EBP(10682) , ESR1(2099) , ESR2(2100) , ESRRA(2101)
rat ... Ar(24208) , Esr1(24890)
Looking for similar products? Visit Product Comparison Guide
General description
This nonsteroidal triphenylethylene derivative suppresses tumor growth and has been reported to be a carcinogen and teratogen in animals. Additionally, TC may also protect bone from estrogen-deficiency bone loss, lower plasma cholesterol, and have been implicated in liver carcinogenesis in rats. Other actions of TC include the reduction of plasma levels of insulin-like growth factor, induction of cells surrounding cancer cells to secrete transforming growth factor β, and inhibition of membrane lipid peroxidation, likely by decreasing membrane fluidity.
Application
- in the MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) assay and crystal violet staining assay to access cell viability
- to induce Dicer ablation in Dicer-conditional mice, to study the effects of Dicer deficiency on adipocyte differentiation and function.
- to study its biological effects on the glucocorticoid (GC)-resistant cell line Jurkat, derived from acute lymphoblastic leukemia of T lineage (T-ALL)
- to study the physiological effects of tamoxifen treatment on the hypothalamus-POA mediated by estrogen receptor alpha (ERα) in mice
- to induce Cre recombinase activity for gene manipulation.
- to facilitate the recombination of the ect2flox allele in mouse organs
- to study its effect on lipopolysaccharide (LPS)-induced microglial activation
Biochem/physiol Actions
Features and Benefits
Preparation Note
Storage and Stability
Other Notes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1A - Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We presents an article on Autophagy in Cancer Promotes Therapeutic Resistance
Related Content
Interrogation of protein pathways is a foundational part of drug discovery research and drug development. Explore more about protein pathway analysis, including chemical library screening, investigating protein and enzyme activity, and modulating protein pathways by using small molecules.
Discover Bioactive Small Molecules for ADME/Tox
Chromatograms
application for HPLC, application for SPEOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service