G1513
Glycerol formal
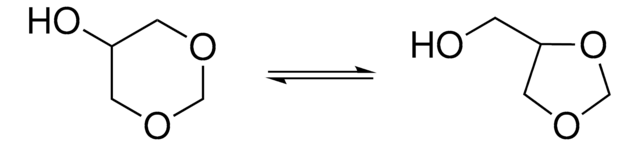
47-67% 5-hydroxy-1,3-dioxane basis (GC), 33-53% 4-hydroxymethyl-1,3-dioxolane basis (GC)
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H8O3
CAS Number:
Molecular Weight:
104.10
Beilstein:
103206
MDL number:
UNSPSC Code:
50161901
PubChem Substance ID:
NACRES:
NA.25
Recommended Products
Quality Level
contains
~0.02% 2,6-di-tert-butyl-4-methylphenol as stabilizer
composition
5-hydroxy-1,3-dioxane, 47-67% GC
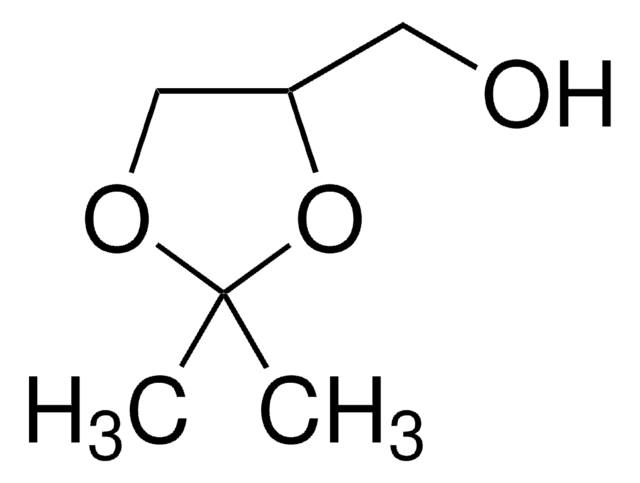
4-hydroxymethyl-1,3-dioxolane, 33-53% GC
refractive index
n20/D 1.451 (lit.)
bp
192-193 °C (lit.)
density
1.203 g/mL at 25 °C (lit.)
SMILES string
OCC1COCO1.OC2COCOC2
InChI
1S/2C4H8O3/c5-4-1-6-3-7-2-4;5-1-4-2-6-3-7-4/h2*4-5H,1-3H2
InChI key
JIUMSISXCKWZTA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Glycerol formal (GlyF) includes isomers, namely, 4-hydroxymethyl-1,3-dioxolane and 5-hydroxy-1,3-dioxane. It is a glycerol synthon with a combination of two 5- and 6-membered ring isomers.
Application
Glycerol formal is used to to solubilize water-insoluble compounds for subsequent aqueous dilution. It has been used as a chemical and dye emulsifier and as a cosolvent for drug delivery. Glycerol formal was used as a vehicle for antibiotic delivery in rats.
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Properties of fatty acid glycerol formal ester (FAGE) for use as a component in blends for diesel engines
Lapuerta M, et al.
Biomass and Bioenergy, 130-140 (2015)
Sarah Nickolls et al.
Advances in pharmacological sciences, 2011, 608912-608912 (2011-12-14)
GABA(A) receptors containing α2/3 subunits are current targets in the battle to develop new pain medications, as they are expressed in the spinal cord where increasing inhibitory drive should result in analgesia. However, this approach is prone to a range
Kazuo Hotta et al.
Anticancer research, 35(9), 4681-4689 (2015-08-09)
We investigated whether hepatic multidrug resistance-associated protein 2 (ABCC2) is involved in the hepatobiliary excretion of regorafenib, a novel multi-kinase inhibitor, using Sprague-Dawley (SD) rats and Eisai hyperbilirubinemic rats (EHBR) lacking the efflux transporter ABCC2. The involvement of organic anion-transporting
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service