Recommended Products
Quality Level
product line
NovaSyn® TG
Novabiochem®
form
beads
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
manufacturer/tradename
Novabiochem®
application(s)
peptide synthesis
functional group
alcohol
storage temp.
2-8°C
Related Categories
General description
Associated Protocols and Technical Articles
Protocols for Loading of Peptide Synthesis Resins
Literature references
[1] E. Bayer, et al. in ′Peptides, Chemistry, Structure & Biology, Proc. 13th American Peptide Symposium′, R. S. Hodges & J. A. Smith(Eds), ESCOM, Leiden, 1994, pp. 156.
[2] J. M. J. Frechet, et al. (1975) Tetrahedron Lett., 3055.
[3] T. M. Fyles, et al. (1976) Can. J. Chem., 54,935.
[4] A. v. Vliet, et al. in ′Innovation & Perspectives in Solid Phase Synthesis, 2nd International Symposium′, R. Epton (Eds), Intercept UKLtd., Andover, 1992, pp. 475.
[5] A. v. Vliet, et al. in ′Peptides 1992, Proc. 22nd European Peptide Symposium′, C. H. Schneider & A. N. Eberle (Eds), ESCOM, Leiden,1993, pp. 279.
[6] G. Grübler, et al. in ′Innovation & Perspectives in Solid Phase Synthesis, 3rd International Symposium′, R. Epton (Eds), Mayflower Worldwide Ltd., Birmingham, 1994, pp. 517.
[7] R. Bollhagen, et al. (1994) J. Chem. Soc., Chem. Commun., 2559.
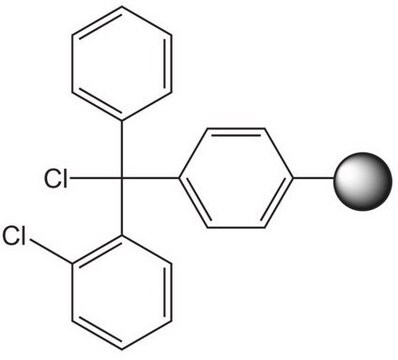
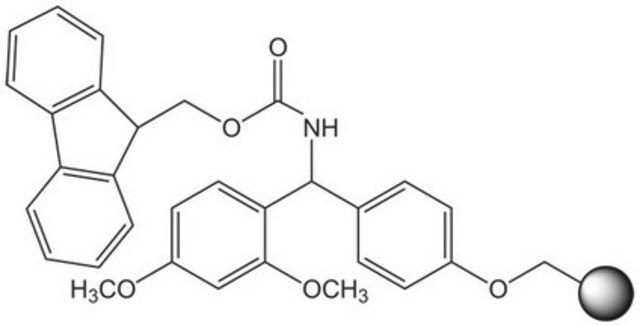
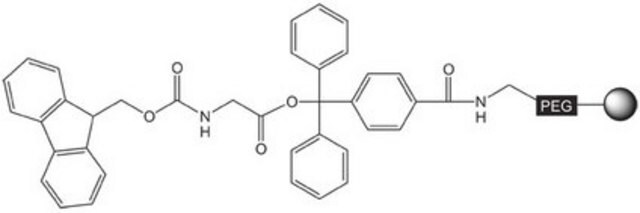
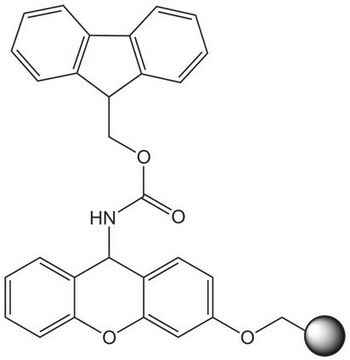
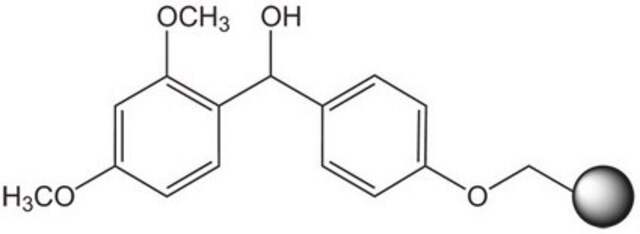
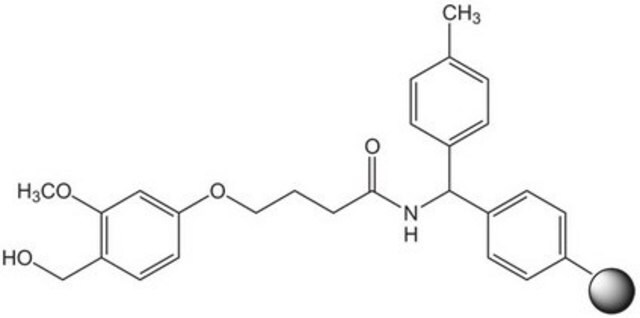
Linkage
Analysis Note
Appearance of substance (visual): beads
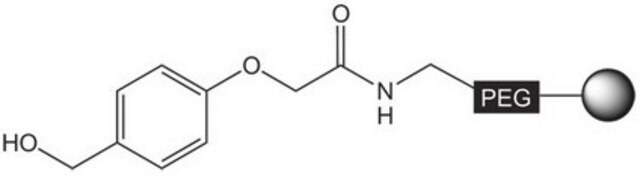
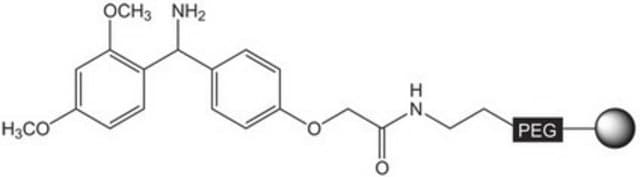
Loading (determined from the substitution of the Fmoc-Leu loaded resin): 0.15 - 0.30 mmol/g
Swelling Volume (in CH₂Cl₂): lot specific result
The polymer matrix is NovaSyn TG resin (90 µm).
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Novabiochem® has one of the most extensive ranges of linkers and derivatized resins for Fmoc solid phase peptide synthesis. These resins have varied properties with special protocols for loading and cleaving.
Protocols
Overcome challenges in synthesis and disulfide bond formation with protocols for Fmoc solid-phase peptide synthesis of peptides with cysteine and methionine.
Review methods and resins for attaching amino acids and peptides, including Merrifield, trityl-based, and hydroxymethyl-functionalized resins. Resin-immobilized peptides can be used for various downstream applications.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service